Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 5.3 E–Z naming system-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.3 E–Z naming system- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.3 E–Z naming system- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
5.3 understand the E–Z naming system for geometric isomers and why it is necessary to use this when the cis- and trans- naming system breaks down
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
5.3 E–Z Naming System for Geometric Isomerism
The E–Z naming system is used to describe geometric isomers when the cis–trans system is not applicable. It provides a more general and systematic way of naming isomers based on the priority of substituents.
Why Cis–Trans Naming Breaks Down
The cis–trans system only works when there are identical groups attached to each carbon of the double bond.
- Requires a pair of identical substituents.
- Cannot be used when all four groups are different.
Example where cis/trans fails

All substituents are different → cannot define “same” or “opposite” easily.
Therefore, the E–Z system is required.
Principle of E–Z Naming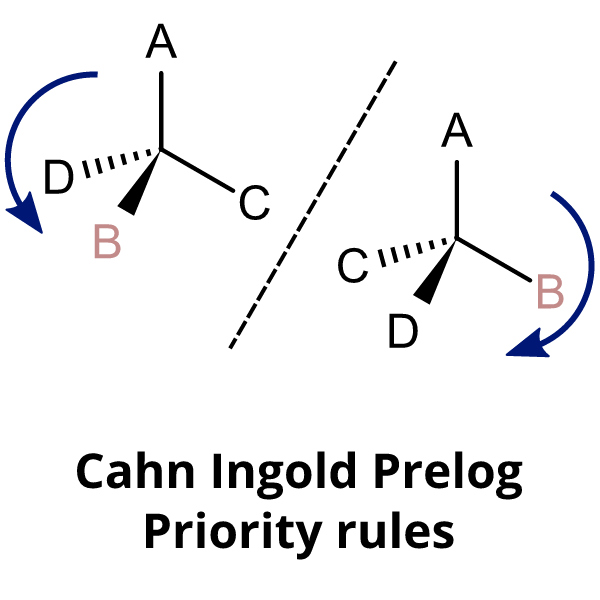
E–Z notation is based on Cahn–Ingold–Prelog (CIP) priority rules.
- Assign priority to substituents on each carbon of the double bond.
- Priority is based on atomic number.
Assigning Priority
- Higher atomic number → higher priority.
- Example: \( \mathrm{Br > Cl > C > H} \)
- If atoms are the same, move to next bonded atoms.
Definition of E and Z

- Z (zusammen)
- Higher priority groups are on the same side of the double bond.
- E (entgegen)
- Higher priority groups are on opposite sides.
Step-by-Step Method
- Step 1: Identify the C=C double bond.
- Step 2: Assign priorities to substituents on each carbon.
- Step 3: Compare positions of highest priority groups.
- Step 4: Same side → Z, opposite side → E.
Example
Consider: \( \mathrm{CH_3CH=CHBr} \)
- Left carbon: \( \mathrm{CH_3 > H} \)
- Right carbon: \( \mathrm{Br > H} \)
- Same side → Z isomer
- Opposite side → E isomer
Key Differences: Cis/Trans vs E/Z
- Cis/trans → limited to simple cases with identical groups.
- E/Z → works for all alkenes with different substituents.
- E/Z is more general and precise.
Summary
- E–Z system uses priority rules to describe geometric isomers.
- Necessary when cis/trans naming is not applicable.
- Z = same side, E = opposite side (based on priority groups).
- Provides a universal method for naming alkenes.
Therefore, the E–Z system ensures accurate and consistent naming of geometric isomers in all cases.
Example 1:
Assign E or Z configuration to \( \mathrm{CH_3CH=C(Cl)Br} \) and justify your answer.
▶️ Answer/Explanation
Left carbon: \( \mathrm{CH_3 > H} \)
Right carbon: \( \mathrm{Br > Cl} \) (Br has higher atomic number)
Compare highest priority groups: \( \mathrm{CH_3} \) and \( \mathrm{Br} \).
If they are on opposite sides → E isomer.
Therefore, the compound is E.
Example 2:
Explain why cis–trans terminology cannot be used for \( \mathrm{CHCl=CHBr} \).
▶️ Answer/Explanation
Each carbon has two different substituents (Cl and H, Br and H).
There are no identical groups to compare positions.
Therefore, cis/trans naming is not applicable.
The E–Z system must be used instead.
