Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 5.4 Reactions of alkenes-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.4 Reactions of alkenes- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.4 Reactions of alkenes- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
5.4 be able to describe the reactions of alkenes, limited to:
i the addition of hydrogen, using a nickel catalyst, to form an alkane
ii the addition of halogens to produce a di-substituted halogenoalkane
iii the addition of hydrogen halides to produce mono-substituted halogenoalkanes
iv the addition of steam, in the presence of an acid catalyst, to produce alcohols
v oxidation of the double bond by acidified potassium manganate(VII) to produce a diol
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
5.4 (i) Addition of Hydrogen (Hydrogenation)
Alkenes undergo addition reactions due to the presence of the electron-rich π bond. One important reaction is hydrogenation, where hydrogen is added across the double bond to form an alkane.
General Reaction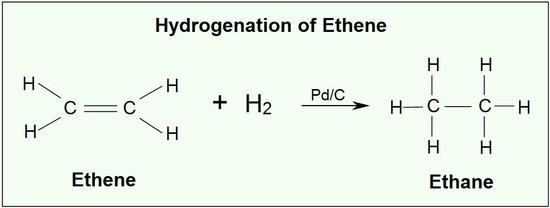
\( \mathrm{C=C + H_2 \rightarrow C–C} \)
Example
- \( \mathrm{CH_2=CH_2 + H_2 \rightarrow CH_3CH_3} \)
Conditions
- Nickel catalyst (\( \mathrm{Ni} \))
- Temperature ≈ 150°C
Explanation
- The π bond breaks.
- Two hydrogen atoms add across the double bond.
- A saturated alkane is formed.
Therefore, hydrogenation converts unsaturated alkenes into saturated alkanes.
5.4 (ii) Addition of Halogens
Alkenes react with halogens such as \( \mathrm{Br_2} \) or \( \mathrm{Cl_2} \) via addition reactions to form di-substituted halogenoalkanes.
General Reaction
\( \mathrm{C=C + X_2 \rightarrow X–C–C–X} \)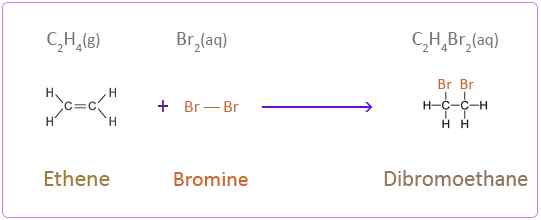
Example
- \( \mathrm{CH_2=CH_2 + Br_2 \rightarrow CH_2BrCH_2Br} \)
Conditions
- Room temperature
- No catalyst required
Observation
- Bromine water changes from orange to colourless.
This is a test for unsaturation.
Explanation
- The π bond breaks.
- Each carbon forms a bond with a halogen atom.
- A vicinal dihalogenoalkane is formed.
Therefore, halogen addition confirms the presence of a double bond and forms saturated products.
Key Comparison
- Hydrogenation → forms alkane (adds \( \mathrm{H_2} \)).
- Halogenation → forms dihalogenoalkane (adds \( \mathrm{X_2} \)).
- Both involve breaking of π bond.
Therefore, these reactions demonstrate the high reactivity of alkenes compared to alkanes.
Example 1 :
An alkene \( \mathrm{C_4H_8} \) reacts with hydrogen to form a single product. Deduce the structure of the alkene and write the equation.
▶️ Answer/Explanation
Hydrogenation produces an alkane \( \mathrm{C_4H_{10}} \).
Possible alkene: but-1-ene or but-2-ene.
Both give butane on hydrogenation:
\( \mathrm{CH_3CH_2CH=CH_2 + H_2 \rightarrow CH_3CH_2CH_2CH_3} \)
Therefore, the product is butane regardless of position of double bond.
Example 2:
An alkene decolourises bromine water to form \( \mathrm{C_3H_6Br_2} \). Deduce the structure of the alkene and explain the reaction.
▶️ Answer/Explanation
Product has 3 carbons → original alkene is \( \mathrm{C_3H_6} \) (propene).
Reaction:
\( \mathrm{CH_3CH=CH_2 + Br_2 \rightarrow CH_3CHBrCH_2Br} \)
The π bond breaks and two bromine atoms add across the double bond.
Therefore, a dibromoalkane is formed and bromine water is decolourised.
(iii) Addition of Hydrogen Halides (HX)
Alkenes react with hydrogen halides (e.g. \( \mathrm{HCl} \), \( \mathrm{HBr} \)) to form mono-substituted halogenoalkanes.
General Reaction 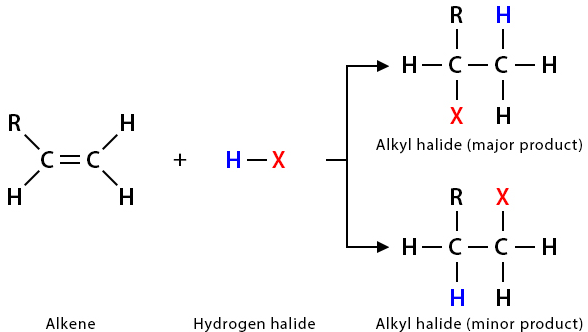
\( \mathrm{C=C + HX \rightarrow C–C} \) (one H and one X added)
Example
- \( \mathrm{CH_3CH=CH_2 + HBr \rightarrow CH_3CHBrCH_3} \)
Mechanism Feature (Important)
- Reaction follows Markovnikov’s rule.
- Hydrogen adds to the carbon with more hydrogens already attached.
- Halogen adds to the more substituted carbon.
Therefore, the major product is the more stable carbocation-derived product.
5.4 (iv) Addition of Steam (Hydration)
Alkenes react with steam to form alcohols in the presence of an acid catalyst.
General Reaction
\( \mathrm{C=C + H_2O \rightarrow alcohol} \) 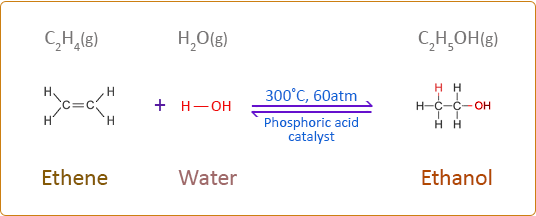
Example
- \( \mathrm{CH_2=CH_2 + H_2O \rightarrow CH_3CH_2OH} \)
Conditions
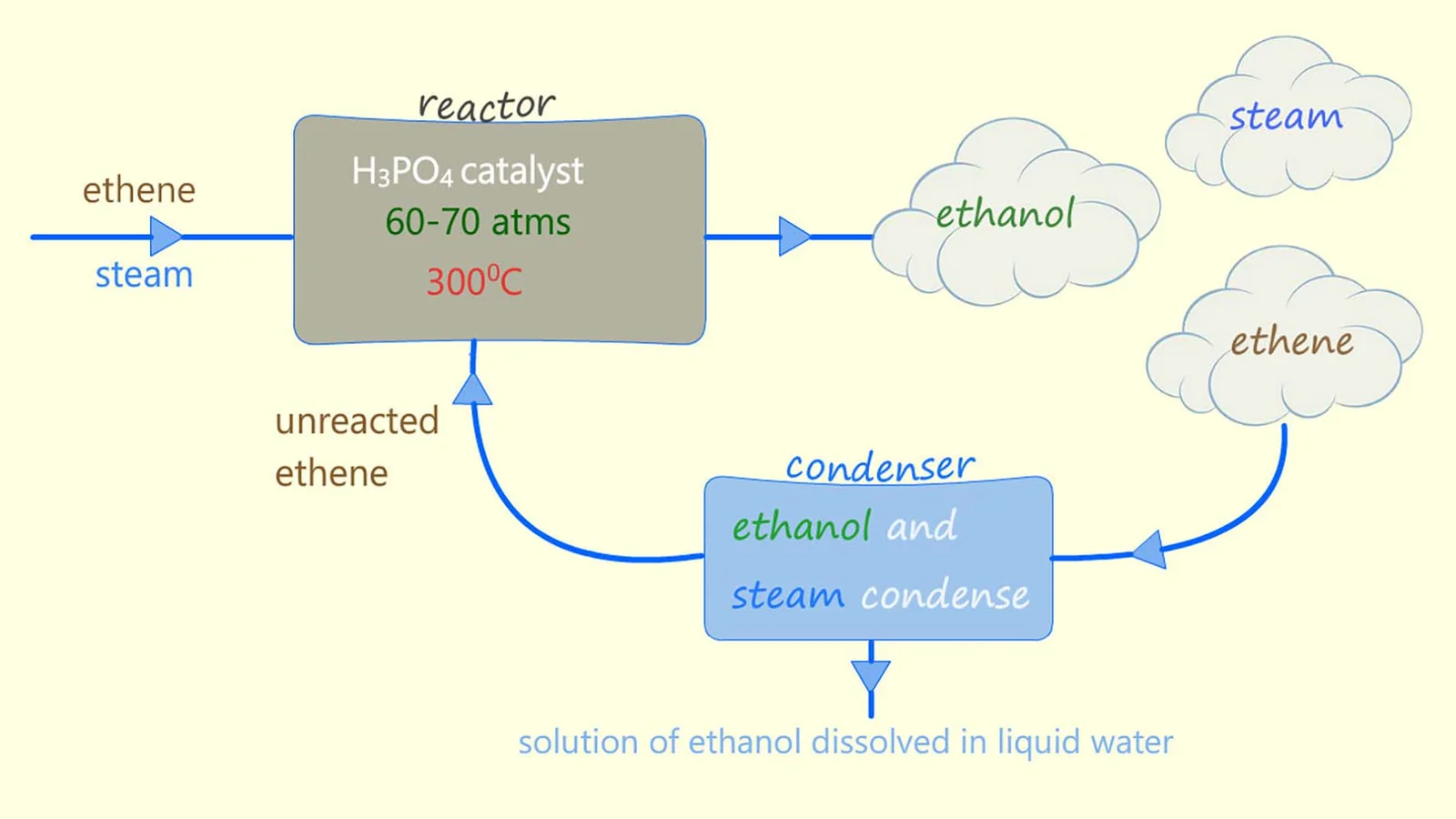
- Phosphoric acid catalyst (\( \mathrm{H_3PO_4} \))
- High temperature (~300°C)
- High pressure (~60–70 atm)
Mechanism Feature
- Also follows Markovnikov’s rule.
Therefore, hydration produces alcohols with predictable orientation.
5.4 (v) Oxidation with Acidified Potassium Manganate(VII)
Alkenes are oxidised by acidified potassium manganate(VII) (\( \mathrm{KMnO_4} \)) to form diols (compounds with two –OH groups).
General Reaction
\( \mathrm{C=C \rightarrow HO–C–C–OH} \)
Example
- \( \mathrm{CH_2=CH_2 \xrightarrow{KMnO_4/H^+} HOCH_2CH_2OH} \)
Conditions
- Cold, dilute, acidified \( \mathrm{KMnO_4} \)
Observation
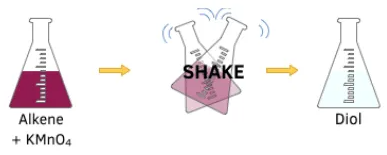
- Purple solution of \( \mathrm{KMnO_4} \) decolourises.
Explanation
- The π bond is broken.
- Two hydroxyl groups (–OH) are added across the double bond.
- A vicinal diol is formed.
Therefore, this reaction is used as a test for unsaturation and produces diols.
Therefore, alkenes undergo a wide range of addition reactions due to their reactive double bond.
Example 1:
Predict the major product of the reaction between propene and hydrogen chloride and explain your answer.
▶️ Answer/Explanation
Reaction follows Markovnikov’s rule.
Hydrogen adds to the carbon with more hydrogens.
Chlorine adds to the more substituted carbon.
Product:
\( \mathrm{CH_3CH=CH_2 + HCl \rightarrow CH_3CHClCH_3} \)
Therefore, 2-chloropropane is the major product.
Example 2 :
An alkene \( \mathrm{C_4H_8} \) reacts with cold, dilute acidified \( \mathrm{KMnO_4} \) to form a diol. Deduce the structure of the product and explain the observation.
▶️ Answer/Explanation
The double bond is oxidised to form a diol.
Example alkene: but-2-ene.
Product:
\( \mathrm{CH_3CH=CHCH_3 \rightarrow CH_3CHOHCHOHCH_3} \)
Purple \( \mathrm{KMnO_4} \) is reduced and decolourises.
Therefore, a vicinal diol is formed and the colour change confirms oxidation.
