Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 5.5 Test for C=C-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.5 Test for C=C- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.5 Test for C=C- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
5.5 know the qualitative test for a C=C double bond using bromine or bromine water
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
5.5 Qualitative Test for a C=C Double Bond
A simple qualitative test for the presence of a carbon–carbon double bond (C=C) is the use of bromine or bromine water. This test is used to distinguish between saturated and unsaturated hydrocarbons.
Reagent
- Bromine water (\( \mathrm{Br_2(aq)} \))
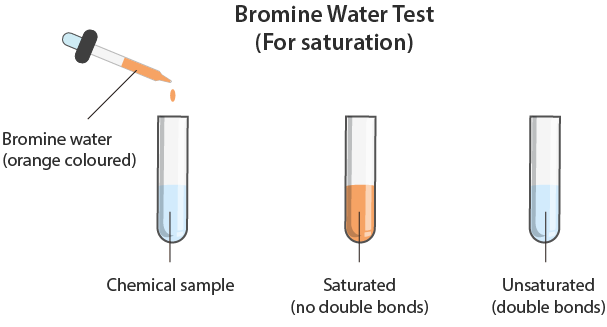
- Orange/brown colour
Procedure
- Add bromine water to the organic compound.
- Shake the mixture at room temperature.
Observation
- Alkene (unsaturated) → bromine water is decolourised (orange → colourless)
- Alkane (saturated) → no colour change
Explanation
- Alkenes undergo addition reaction with bromine.
- The π bond breaks.
- Bromine adds across the double bond.
- A colourless dibromoalkane is formed.
Example
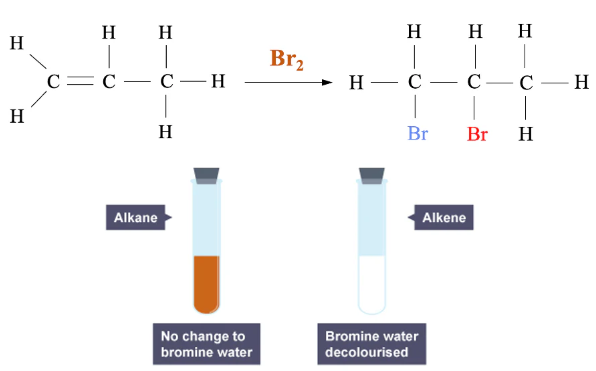
Why Alkanes Do Not React
- Alkanes do not have a π bond.
- No addition reaction occurs under these conditions.
- Bromine colour remains unchanged.
Key Exam Points
- Test for unsaturation (C=C bond).
- Positive result → decolourisation of bromine water.
- Reaction occurs at room temperature (no UV needed).
- Do not confuse with alkane + halogen (needs UV).
Therefore, bromine water provides a simple and effective qualitative test for unsaturation.
Example 1 :
A hydrocarbon \( \mathrm{C_4H_8} \) decolourises bromine water instantly at room temperature. Explain what this indicates about its structure.
▶️ Answer/Explanation
Decolourisation indicates presence of a C=C double bond.
Therefore, the compound is an alkene (unsaturated).
The π bond reacts with bromine via addition.
Hence, it is not an alkane.
Example 2 :
A compound does not decolourise bromine water but reacts with chlorine under UV light. Deduce the type of compound and explain your reasoning.
▶️ Answer/Explanation
No reaction with bromine water → no C=C bond.
Reaction with chlorine under UV indicates free radical substitution.
This is characteristic of alkanes.
Therefore, the compound is a saturated alkane.
