Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 5.6 Electrophilic addition mechanism-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.6 Electrophilic addition mechanism- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.6 Electrophilic addition mechanism- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
5.6 be able to describe the mechanism (including diagrams), giving evidence where possible, of:
i the electrophilic addition of bromine and hydrogen bromide to ethene
ii the electrophilic addition of hydrogen bromide to propene
Use of the curly arrow notation is expected – the curly arrows should start from either a bond or from a lone pair of electrons.
Knowledge of the relative stability of primary, secondary and tertiary carbocation intermediates is expected.
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
5.6 (i) Electrophilic Addition to Ethene
Alkenes such as ethene undergo electrophilic addition because the π bond is electron-rich and can attract electrophiles. The mechanism involves curly arrows showing movement of electron pairs.
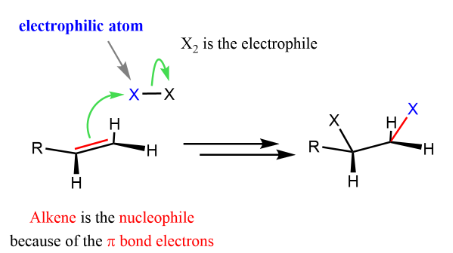
Reaction 1: Addition of Bromine to Ethene
Overall Equation
\( \mathrm{CH_2=CH_2 + Br_2 \rightarrow CH_2BrCH_2Br} \)
Step 1: Formation of Electrophile
- The π bond in ethene polarises the \( \mathrm{Br_2} \) molecule.
- One Br becomes δ⁺ (electrophile), the other δ⁻.
Step 2: Attack by π Electrons
- Curly arrow from π bond → Brδ⁺.
- Br–Br bond breaks (curly arrow to Br).
- Forms a carbocation intermediate (or bromonium ion conceptually).
Step 3: Nucleophilic Attack
- \( \mathrm{Br^-} \) attacks the carbocation.
- Curly arrow from lone pair on \( \mathrm{Br^-} \) → carbon.
- Forms \( \mathrm{CH_2BrCH_2Br} \).
Therefore, bromine adds across the double bond forming a dibromoalkane.
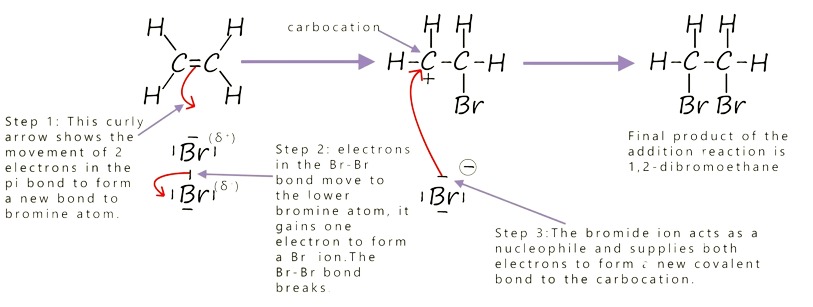
Reaction 2: Addition of Hydrogen Bromide to Ethene
Overall Equation
\( \mathrm{CH_2=CH_2 + HBr \rightarrow CH_3CH_2Br} \)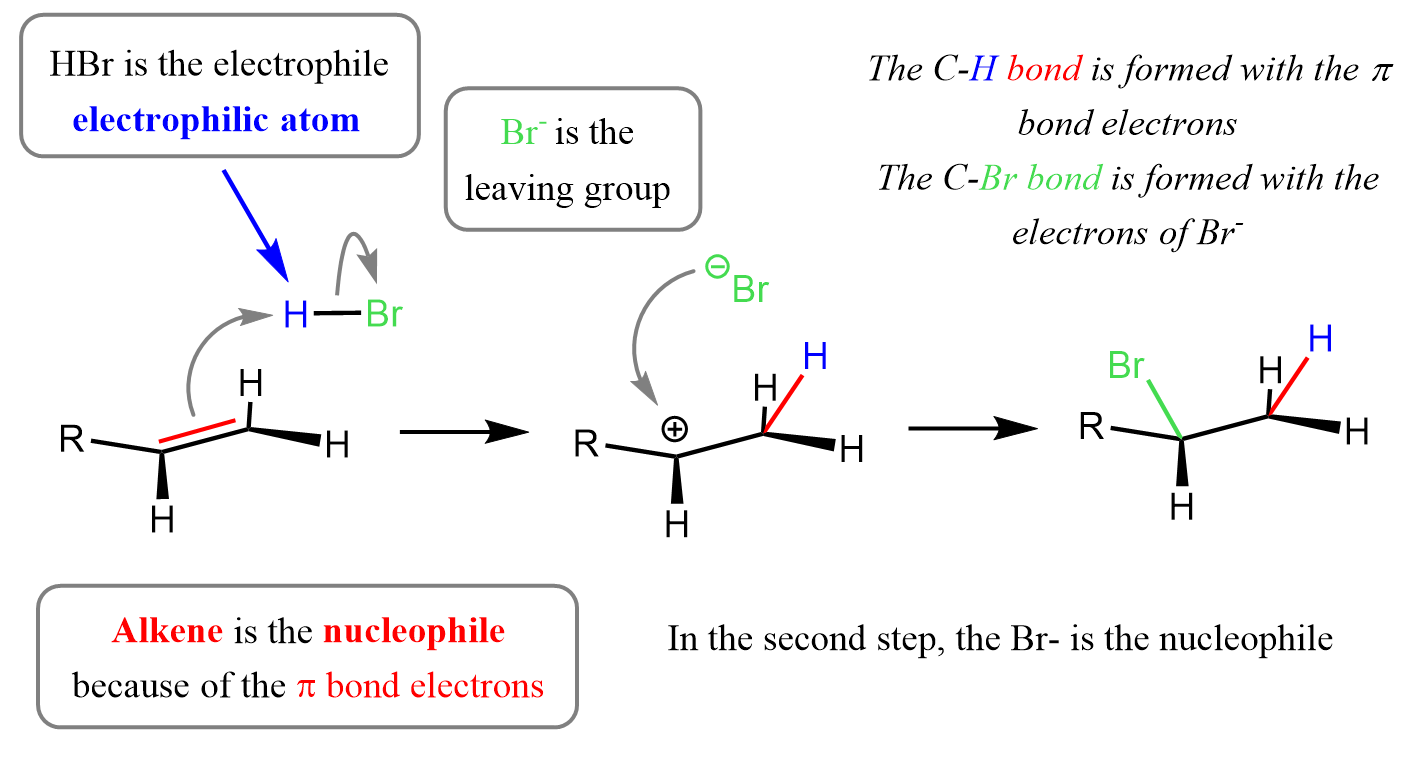
Step 1: Electrophilic Attack (Protonation)
- \( \mathrm{HBr} \) is polar: Hδ⁺–Brδ⁻.
- Curly arrow from π bond → H⁺.
- H–Br bond breaks → \( \mathrm{Br^-} \).
- Forms a carbocation intermediate.
Step 2: Nucleophilic Attack
- \( \mathrm{Br^-} \) attacks the carbocation.
- Curly arrow from lone pair → carbon.
- Forms bromoethane.
Key Features of Electrophilic Addition
- π bond acts as a source of electrons.
- Electrophile is attacked first.
- Carbocation intermediate is formed.
- Nucleophile completes the reaction.
Evidence for the Mechanism
- Rapid reaction supports a low activation energy pathway.
- Formation of intermediates explains observed products.
- Reaction with bromine explains decolourisation of bromine water.
Therefore, electrophilic addition explains the reactivity of alkenes and formation of addition products.
Example 1:
Using curly arrow notation, explain why bromine acts as an electrophile in its reaction with ethene.
▶️ Answer/Explanation
The π bond in ethene is electron-rich.
It induces a dipole in \( \mathrm{Br_2} \), making one Br atom δ⁺.
A curly arrow from the π bond attacks Brδ⁺.
Therefore, bromine behaves as an electrophile.
Example 2 :
Explain why a carbocation intermediate is formed during the reaction of ethene with HBr.
▶️ Answer/Explanation
The π bond donates electrons to H⁺.
This forms a new C–H bond.
The other carbon is left electron-deficient.
Therefore, a positively charged carbocation intermediate is formed.
5.6 (ii) Electrophilic Addition of HBr to Propene
The electrophilic addition of hydrogen bromide to propene demonstrates regioselectivity and the importance of carbocation stability.
Overall Reaction
\( \mathrm{CH_3CH=CH_2 + HBr \rightarrow CH_3CHBrCH_3} \) (major product)
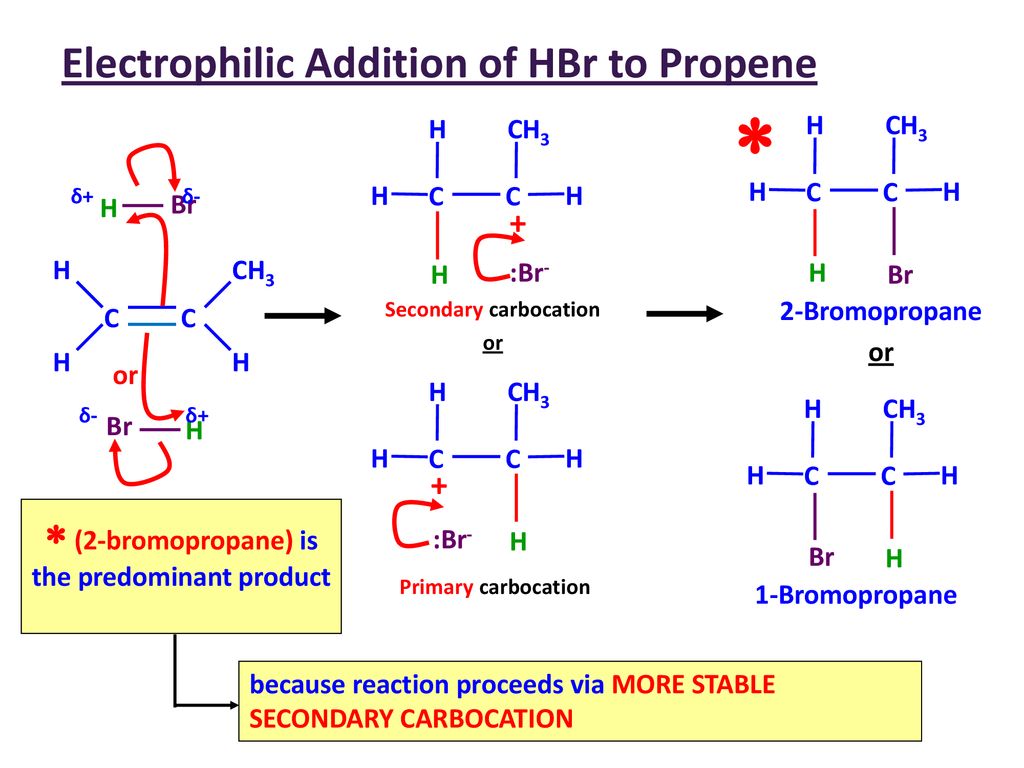
Minor product may also form:
\( \mathrm{CH_3CH_2CH_2Br} \)
Step 1: Electrophilic Attack (Formation of Carbocation)
- \( \mathrm{HBr} \) is polar: Hδ⁺–Brδ⁻.
- Curly arrow from π bond → H⁺.
- H–Br bond breaks → \( \mathrm{Br^-} \).
Two Possible Carbocations
- Primary carbocation:
- \( \mathrm{CH_3CH_2CH_2^+} \) (less stable)
- Secondary carbocation:
- \( \mathrm{CH_3CH^+CH_3} \) (more stable)
The secondary carbocation is favoured because it is more stable.
Reason for Carbocation Stability
- Alkyl groups donate electron density (+I effect).
- This stabilises the positive charge.
Order of stability:
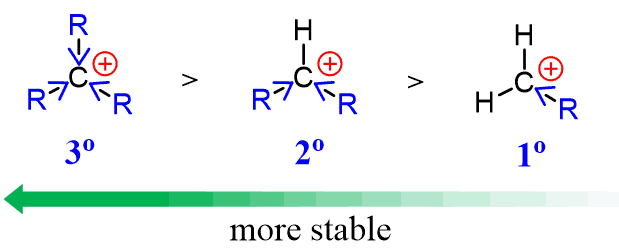
tertiary > secondary > primary
Step 2: Nucleophilic Attack
- \( \mathrm{Br^-} \) attacks the carbocation.
- Curly arrow from lone pair → positively charged carbon.
- Forms 2-bromopropane (major product).
Markovnikov’s Rule
- Hydrogen adds to the carbon with more hydrogens.
- Halogen adds to the more substituted carbon.
Therefore, 2-bromopropane is the major product.
Evidence for the Mechanism
- Formation of major and minor products suggests different carbocations.
- Greater yield of major product supports greater stability of secondary carbocation.
Therefore, carbocation stability determines the outcome of the reaction.
Example 1:
Explain why 2-bromopropane is formed as the major product when propene reacts with HBr.
▶️ Answer/Explanation
The reaction proceeds via a carbocation intermediate.
The secondary carbocation is more stable than the primary carbocation.
Therefore, it forms preferentially.
Bromide ion attacks this carbocation.
Hence, 2-bromopropane is the major product.
Example 2:
Draw the mechanism for the reaction of propene with HBr and explain the formation of both major and minor products.
▶️ Answer/Explanation
Step 1: π bond attacks H⁺ forming a carbocation.
Two carbocations possible: primary and secondary.
Secondary carbocation is more stable → major pathway.
Step 2: \( \mathrm{Br^-} \) attacks carbocation.
Major product: \( \mathrm{CH_3CHBrCH_3} \).
Minor product forms via less stable primary carbocation.
