Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 5.7 Addition polymerisation-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.7 Addition polymerisation- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.7 Addition polymerisation- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
5.7 be able to describe the addition polymerisation of alkenes and draw the repeat unit given the monomer, and vice versa
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
5.7 Addition Polymerisation of Alkenes
Addition polymerisation is the process in which many small alkene molecules (monomers) join together to form a large molecule called a polymer.
Key Concept
The π bond in the alkene breaks, allowing monomers to link together into a long chain.
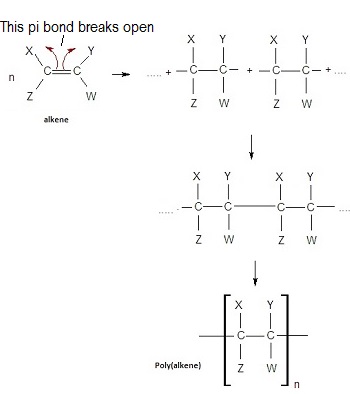
- No other products are formed → addition polymerisation.
- Produces a saturated polymer.
General Reaction
\( \mathrm{n(CH_2=CHX) \rightarrow [-CH_2-CHX-]_n} \)
- \( n \) = large number of repeating units
Example 1: Ethene → Poly(ethene)
\( \mathrm{n(CH_2=CH_2) \rightarrow [-CH_2-CH_2-]_n} \)
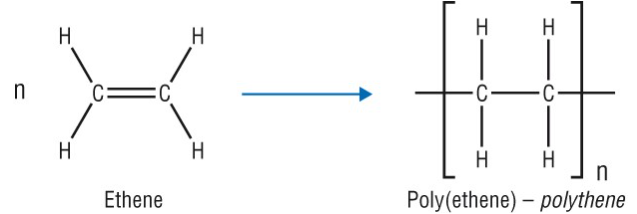
- Double bond breaks.
- Forms long chain of repeating units.
Example 2: Chloroethene → PVC
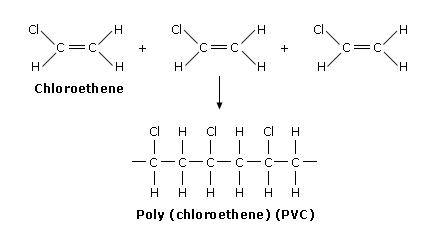
\( \mathrm{n(CH_2=CHCl) \rightarrow [-CH_2-CHCl-]_n} \)
Drawing the Repeat Unit from the Monomer
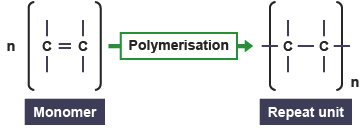
- Step 1: Identify the C=C double bond.
- Step 2: Break the double bond → form single bond.
- Step 3: Add bonds extending from both carbons.
- Step 4: Place structure in brackets with subscript \( n \).
Drawing the Monomer from the Repeat Unit
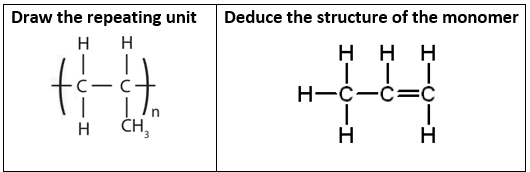
- Step 1: Identify the repeating unit.
- Step 2: Insert a double bond between the two carbons.
- Step 3: Remove extension bonds.
Key Features of Addition Polymerisation
- Only occurs in unsaturated monomers (alkenes).
- Involves breaking of π bond.
- Produces long-chain saturated polymers.
- No small molecules eliminated.
Therefore, understanding the relationship between monomers and repeat units is essential for polymer chemistry.
Example 1 :
The monomer \( \mathrm{CH_2=CHCH_3} \) undergoes addition polymerisation. Draw the repeat unit of the polymer.
▶️ Answer/Explanation
Break the C=C double bond.
Form single bonds and extend chain:
\( \mathrm{[-CH_2-CH(CH_3)-]_n} \)
Therefore, the polymer is poly(propene).
Example 2:
A polymer has the repeat unit \( \mathrm{[-CH_2-CHBr-]_n} \). Deduce the monomer and explain your reasoning.
▶️ Answer/Explanation
Identify the two carbon atoms in the repeat unit.
Reintroduce a double bond between them.
Monomer:
\( \mathrm{CH_2=CHBr} \)
Therefore, the monomer is bromoethene.
