Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 1 - 5.8 Polymer disposal and environmental issues-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.8 Polymer disposal and environmental issues- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 1 – 5.8 Polymer disposal and environmental issues- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
5.8 understand how chemists limit the problems caused by polymer disposal by:
i developing biodegradable polymers
ii removing toxic waste gases produced by the incineration of polymers
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
5.8 Limiting Problems of Polymer Disposal
Disposal of polymers (plastics) causes significant environmental problems because most are non-biodegradable and persist in the environment. Chemists develop methods to reduce these impacts.
(i) Development of Biodegradable Polymers
Biodegradable polymers are designed to break down naturally by the action of microorganisms such as bacteria and fungi.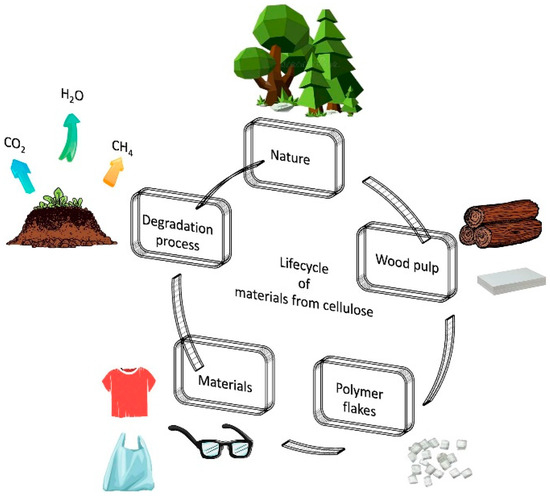
How They Work
- Contain hydrolysable bonds (e.g. ester bonds).
- Water and enzymes break these bonds (hydrolysis).
- Polymer chains break into smaller molecules.
- These are further decomposed into \( \mathrm{CO_2} \), \( \mathrm{H_2O} \), and biomass.
Examples
- Polyesters such as polylactic acid (PLA).
- Starch-based polymers.
Advantages
- Reduce accumulation of plastic waste.
- Lower environmental pollution.
- Can be composted.
Limitations
- May require specific conditions (temperature, moisture).
- Often more expensive to produce.
- May not degrade quickly in landfill conditions.
Therefore, biodegradable polymers reduce long-term waste but are not a complete solution.
(ii) Removal of Toxic Waste Gases from Incineration
Incineration (burning) of polymers is used to reduce waste volume and generate energy, but it produces toxic gases.
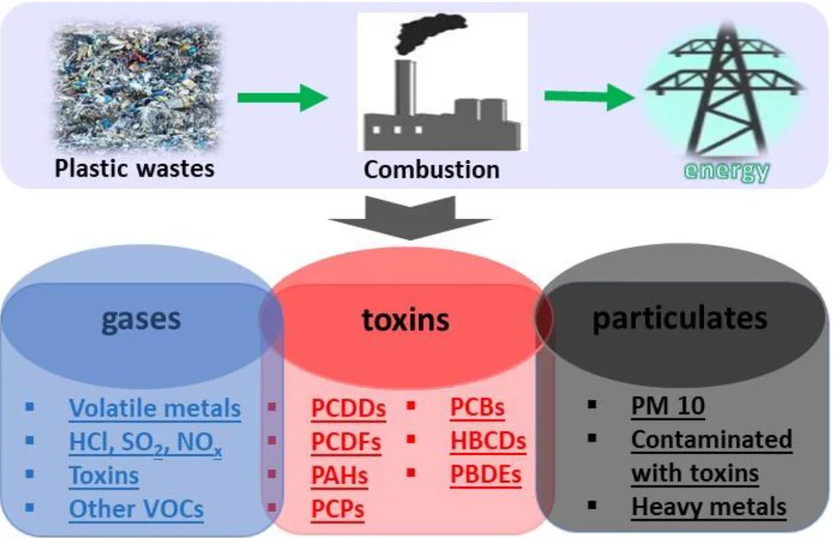
Toxic Gases Produced
- Hydrogen chloride (\( \mathrm{HCl} \)) from PVC.
- Carbon monoxide (\( \mathrm{CO} \)) from incomplete combustion.
- Sulfur dioxide (\( \mathrm{SO_2} \)) and nitrogen oxides (\( \mathrm{NO_x} \)).
- Dioxins (toxic organic compounds).
Methods to Remove Toxic Gases
Scrubbing with alkali:
- Acidic gases such as \( \mathrm{HCl} \) and \( \mathrm{SO_2} \) are neutralised using bases (e.g. \( \mathrm{Ca(OH)_2} \)).
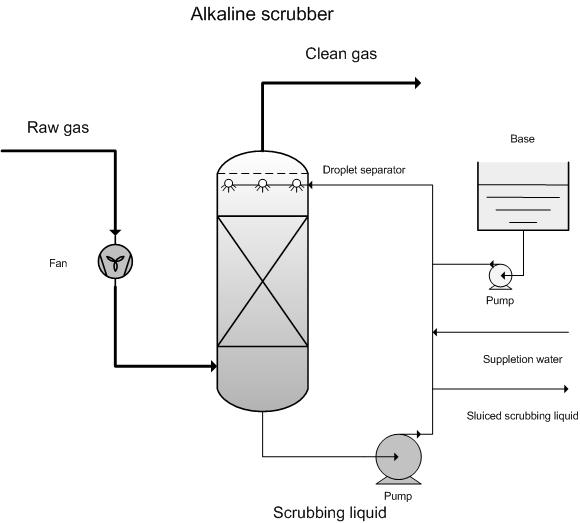
Example: \( \mathrm{HCl + Ca(OH)_2 \rightarrow CaCl_2 + H_2O} \)
Catalytic converters:
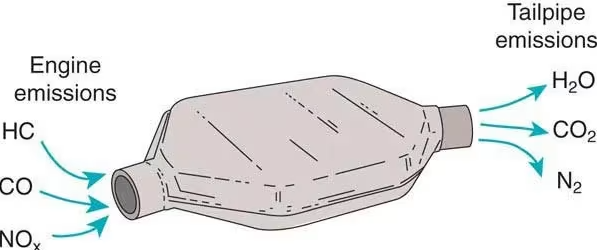
- Convert CO into \( \mathrm{CO_2} \).
- Reduce \( \mathrm{NO_x} \) to \( \mathrm{N_2} \).
High-temperature incineration:
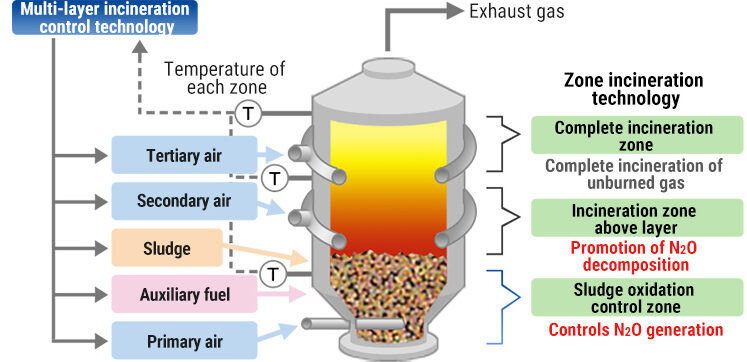
- Ensures complete combustion → reduces CO formation.
Filters and electrostatic precipitators:
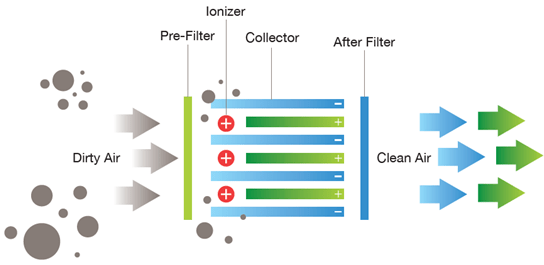
- Remove particulate matter and solid pollutants.
Advantages
- Reduces harmful emissions.
- Makes incineration safer.
- Allows energy recovery from waste.
Therefore, gas treatment systems are essential to minimise environmental damage from burning polymers.
Therefore, chemists use both material design and emission control to manage polymer waste effectively.
Example 1 :
Explain why poly(ethene) is not biodegradable, whereas some polyesters are.
▶️ Answer/Explanation
Poly(ethene) contains only strong C–C and C–H bonds.
These are not easily broken by enzymes or water.
Polyesters contain ester bonds.
These can be hydrolysed by water and enzymes.
Therefore, polyesters are biodegradable but poly(ethene) is not.
Example 2 :
Explain how acidic gases produced during polymer incineration can be removed and why this is necessary.
▶️ Answer/Explanation
Acidic gases such as \( \mathrm{HCl} \) and \( \mathrm{SO_2} \) are neutralised using alkalis such as \( \mathrm{Ca(OH)_2} \).
This forms salts and water.
Removal is necessary because these gases cause acid rain and respiratory problems.
Therefore, scrubbing reduces environmental and health impacts.
