Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.1 Types of reactions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.1 Types of reactions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.1 Types of reactions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.1 be able to classify reactions (including those in Unit 1) as addition, elimination, substitution, oxidation, reduction, hydrolysis or polymerisation
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.1 Classification of Organic Reactions
Organic reactions can be classified into different types based on the overall change occurring in the reactants. Recognising these reaction types is essential for understanding reaction mechanisms and predicting products. These classifications apply across reactions studied in Unit 1 and beyond.
Addition Reactions
An addition reaction is one in which two molecules combine to form a single product, typically involving the breaking of a multiple bond.
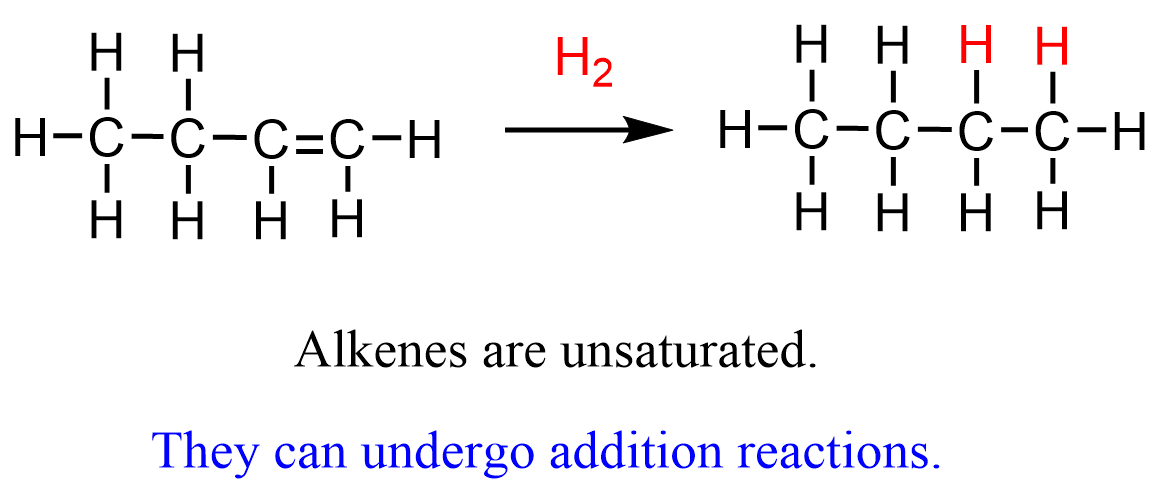
- Occurs mainly in unsaturated compounds such as alkenes.
- The \( \mathrm{C=C} \) double bond is broken and new single bonds are formed.
- Results in a more saturated product.
Example: \( \mathrm{CH_2=CH_2 + H_2 \rightarrow CH_3-CH_3} \)
Substitution Reactions
A substitution reaction is one in which an atom or group in a molecule is replaced by another atom or group.
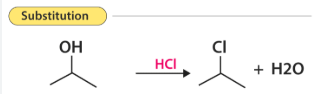
- Common in alkanes and haloalkanes.
- One bond is broken and a new bond is formed.
- The carbon skeleton remains unchanged.
Example: \( \mathrm{CH_4 + Cl_2 \rightarrow CH_3Cl + HCl} \)
Elimination Reactions
An elimination reaction is one in which a small molecule is removed from a larger molecule, resulting in the formation of a multiple bond.
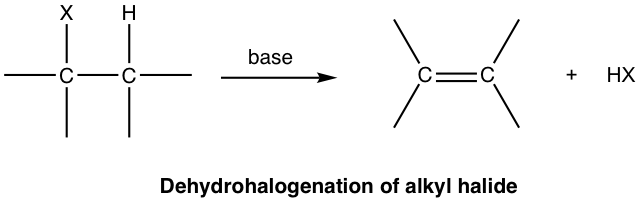
- Often involves removal of \( \mathrm{H_2O} \) or \( \mathrm{HX} \).
- Produces unsaturated compounds such as alkenes.
- Opposite of addition reactions.
Example: \( \mathrm{CH_3CH_2OH \rightarrow CH_2=CH_2 + H_2O} \)
Oxidation Reactions
Oxidation in organic chemistry involves the gain of oxygen and/or loss of hydrogen.
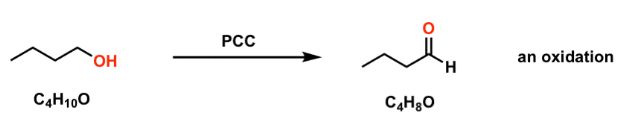
- Increase in number of bonds to oxygen.
- Decrease in number of bonds to hydrogen.
- Often involves oxidising agents such as \( \mathrm{K_2Cr_2O_7} \).
Example: \( \mathrm{CH_3CH_2OH \rightarrow CH_3CHO \rightarrow CH_3COOH} \)
Reduction Reactions
Reduction involves the gain of hydrogen and/or loss of oxygen.

- Opposite of oxidation.
- Often involves hydrogenation reactions.
- Decreases oxidation state of carbon.
Example: \( \mathrm{CH_2=CH_2 + H_2 \rightarrow CH_3CH_3} \)
Hydrolysis Reactions
Hydrolysis is the breaking of a chemical bond by reaction with water.
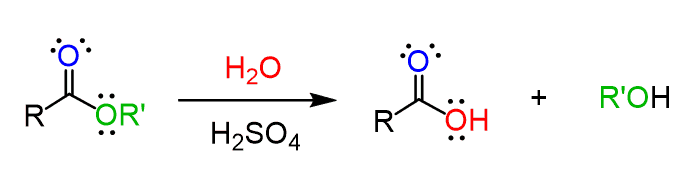
- Water is used to split molecules.
- Common in esters and haloalkanes.
- Produces alcohols, acids, or other functional groups.
Example: \( \mathrm{CH_3CH_2Br + OH^- \rightarrow CH_3CH_2OH + Br^-} \)
Polymerisation Reactions
Polymerisation is the process in which many small molecules (monomers) join together to form a large molecule (polymer).
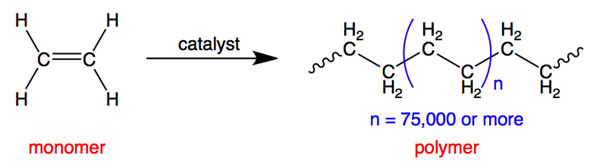
- Occurs in alkenes (addition polymerisation).
- Forms long-chain molecules.
- No small molecule is eliminated in addition polymerisation.
Example: \( \mathrm{nCH_2=CH_2 \rightarrow (-CH_2-CH_2-)_n} \)
Example 1:
Classify the following reaction and justify your answer: \( \mathrm{CH_2=CH_2 + Br_2 \rightarrow CH_2Br-CH_2Br} \)
▶️ Answer/Explanation
This is an addition reaction.
The \( \mathrm{C=C} \) double bond in ethene is broken and two bromine atoms are added across the bond.
Two molecules combine to form a single product without loss of any atoms, which is characteristic of addition reactions.
Example 2:
Classify the following reaction and explain your reasoning: \( \mathrm{CH_3CH_2Br + OH^- \rightarrow CH_3CH_2OH + Br^-} \)
▶️ Answer/Explanation
This is a substitution reaction (specifically nucleophilic substitution).
The bromine atom in the haloalkane is replaced by a hydroxide ion.
One bond is broken and a new bond is formed without changing the carbon skeleton, which is characteristic of substitution reactions.
