Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.10 Hydrolysis rate comparisons-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.10 Hydrolysis rate comparisons- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.10 Hydrolysis rate comparisons- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.10 understand that experimental observations and data can be used to compare the relative rates of hydrolysis of:
i primary, secondary and tertiary structural isomers of a halogenoalkane
ii primary chloro-, bromo- and iodoalkanes using aqueous silver nitrate in ethanol
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.10 Comparing Rates of Hydrolysis of Halogenoalkanes
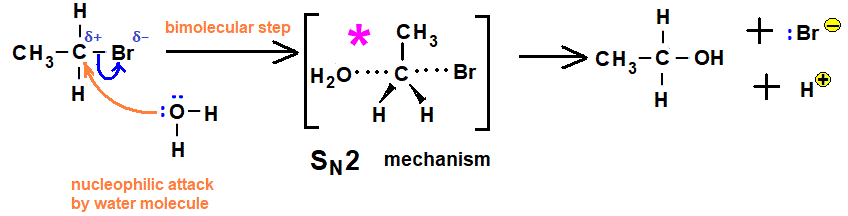
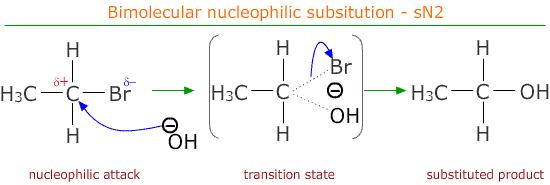
The relative rates of hydrolysis of halogenoalkanes can be determined experimentally using aqueous silver nitrate in ethanol. This reaction involves nucleophilic substitution where water acts as a nucleophile, and the rate can be observed by the formation of a silver halide precipitate. Differences in structure and bond strength influence the rate of hydrolysis.
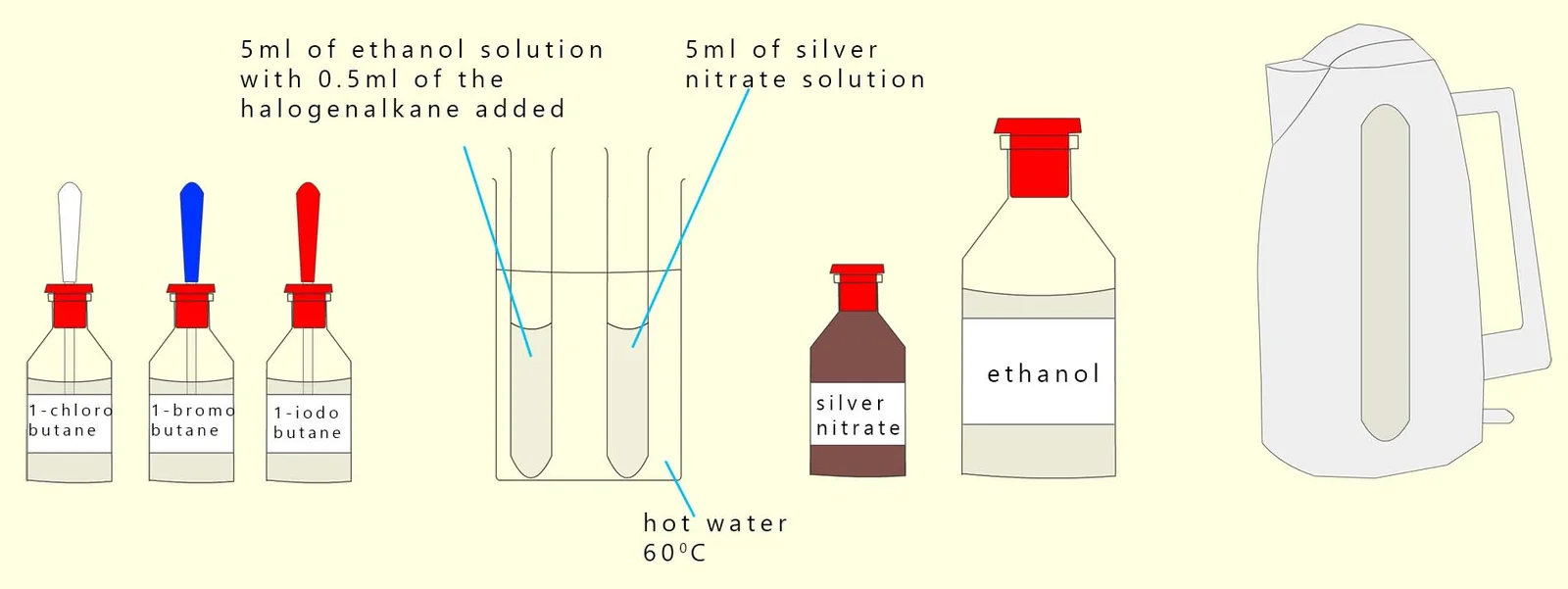
Method Using Aqueous Silver Nitrate in Ethanol
Halogenoalkanes are reacted with aqueous silver nitrate in ethanol, and the time taken for a precipitate to form is measured.
- Water acts as a nucleophile and hydrolyses the halogenoalkane.
- The \( \mathrm{C-X} \) bond breaks heterolytically to release halide ions.
- The halide ions react with \( \mathrm{Ag^+} \) to form a precipitate.
- The rate of precipitate formation indicates the rate of hydrolysis.
(i) Effect of Structure: Primary, Secondary and Tertiary
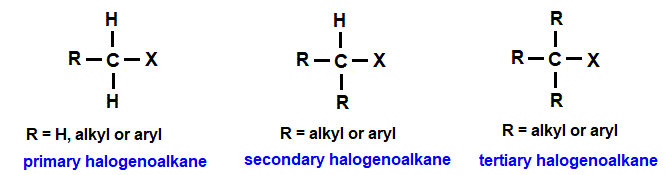
- Tertiary halogenoalkanes hydrolyse fastest.
- Secondary halogenoalkanes hydrolyse at a moderate rate.
- Primary halogenoalkanes hydrolyse slowest.
This trend is explained by the mechanism:
- Tertiary halogenoalkanes form stable carbocations and undergo \( \mathrm{S_N1} \) mechanisms.

- The stability of the carbocation increases the rate of bond breaking.
- Primary halogenoalkanes undergo \( \mathrm{S_N2} \), which is slower due to the need for collision with the nucleophile.

(ii) Effect of Halogen: Cl, Br, I
- Iodoalkanes react fastest.

- Bromoalkanes react at an intermediate rate.
- Chloroalkanes react slowest.
This is due to bond strength:
- The \( \mathrm{C-I} \) bond is weakest and breaks most easily.
- The \( \mathrm{C-Br} \) bond is stronger than \( \mathrm{C-I} \) but weaker than \( \mathrm{C-Cl} \).
- The \( \mathrm{C-Cl} \) bond is strongest, so it breaks most slowly.
Observations
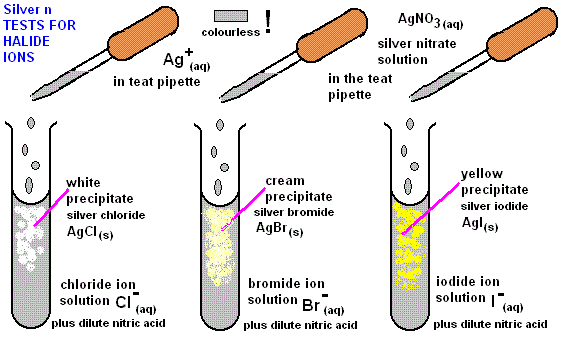
- \( \mathrm{AgCl} \): white precipitate
- \( \mathrm{AgBr} \): cream precipitate
- \( \mathrm{AgI} \): yellow precipitate
Faster formation of precipitate indicates a faster rate of hydrolysis.
Key Ideas
- Rate depends on both bond strength and mechanism.
- Carbocation stability is important for \( \mathrm{S_N1} \) reactions.
- Bond enthalpy determines ease of bond breaking.
- Experimental observations (precipitate formation) allow comparison of rates.
Example 1:
Explain why tertiary halogenoalkanes hydrolyse faster than primary halogenoalkanes.
▶️ Answer/Explanation
Tertiary halogenoalkanes undergo hydrolysis via an \( \mathrm{S_N1} \) mechanism.
This involves formation of a tertiary carbocation, which is stabilised by three alkyl groups through electron-donating effects.
The increased stability lowers the energy required for bond breaking, increasing the rate.
Primary halogenoalkanes do not form stable carbocations and react via the slower \( \mathrm{S_N2} \) mechanism.
Example 2:
Explain why iodoalkanes hydrolyse faster than chloroalkanes using aqueous silver nitrate in ethanol.
▶️ Answer/Explanation
The rate of hydrolysis depends on how easily the \( \mathrm{C-X} \) bond breaks.
The \( \mathrm{C-I} \) bond is weaker than the \( \mathrm{C-Cl} \) bond due to the larger size of iodine and poorer orbital overlap.
Therefore, the \( \mathrm{C-I} \) bond breaks more readily, forming iodide ions faster.
This leads to faster formation of the \( \mathrm{AgI} \) precipitate compared to \( \mathrm{AgCl} \).