Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.11 CORE PRACTICAL 5: Hydrolysis rates-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.11 CORE PRACTICAL 5: Hydrolysis rates- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.11 CORE PRACTICAL 5: Hydrolysis rates- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.11 CORE PRACTICAL 5
Investigation of the rates of hydrolysis of some halogenoalkanes
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.11 Core Practical 5: Investigation of the Rates of Hydrolysis of Halogenoalkanes
The rates of hydrolysis of halogenoalkanes can be investigated experimentally using aqueous silver nitrate in ethanol. This practical allows comparison of how structure and bond type affect the rate of nucleophilic substitution, based on the time taken for a precipitate to form.
Principle of the Experiment
Halogenoalkanes undergo hydrolysis where water acts as a nucleophile, producing alcohols and halide ions, which react with silver ions to form a precipitate.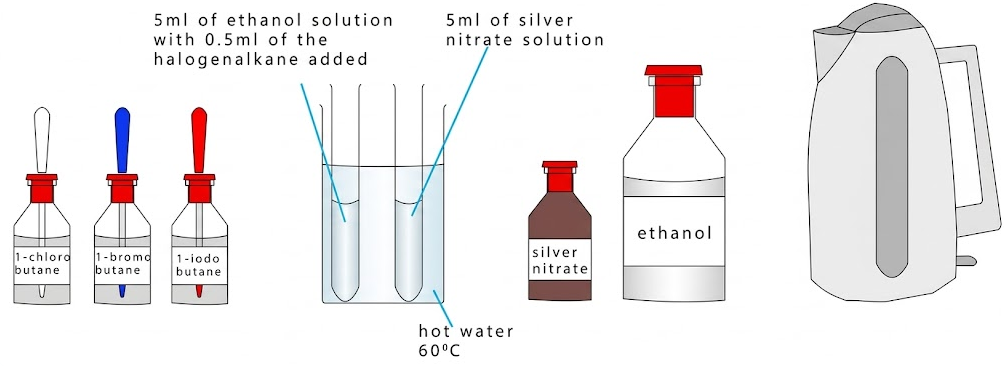
\( \mathbf{R-X + H_2O \rightarrow R-OH + H^+ + X^-} \)
\( \mathbf{Ag^+ + X^- \rightarrow AgX(s)} \)
The rate of formation of the silver halide precipitate is used as a measure of the rate of hydrolysis.
Method
- Add a measured volume of aqueous silver nitrate to a test tube.
- Add ethanol to ensure the halogenoalkane dissolves.
- Place the mixture in a water bath at a constant temperature.
- Add the halogenoalkane and start timing immediately.
- Record the time taken for a precipitate to appear.
Variables
- Independent variable: type of halogenoalkane (structure or halogen).
- Dependent variable: time taken for precipitate formation (rate of hydrolysis).
- Controlled variables: temperature, concentration, volume of reagents.
Observations
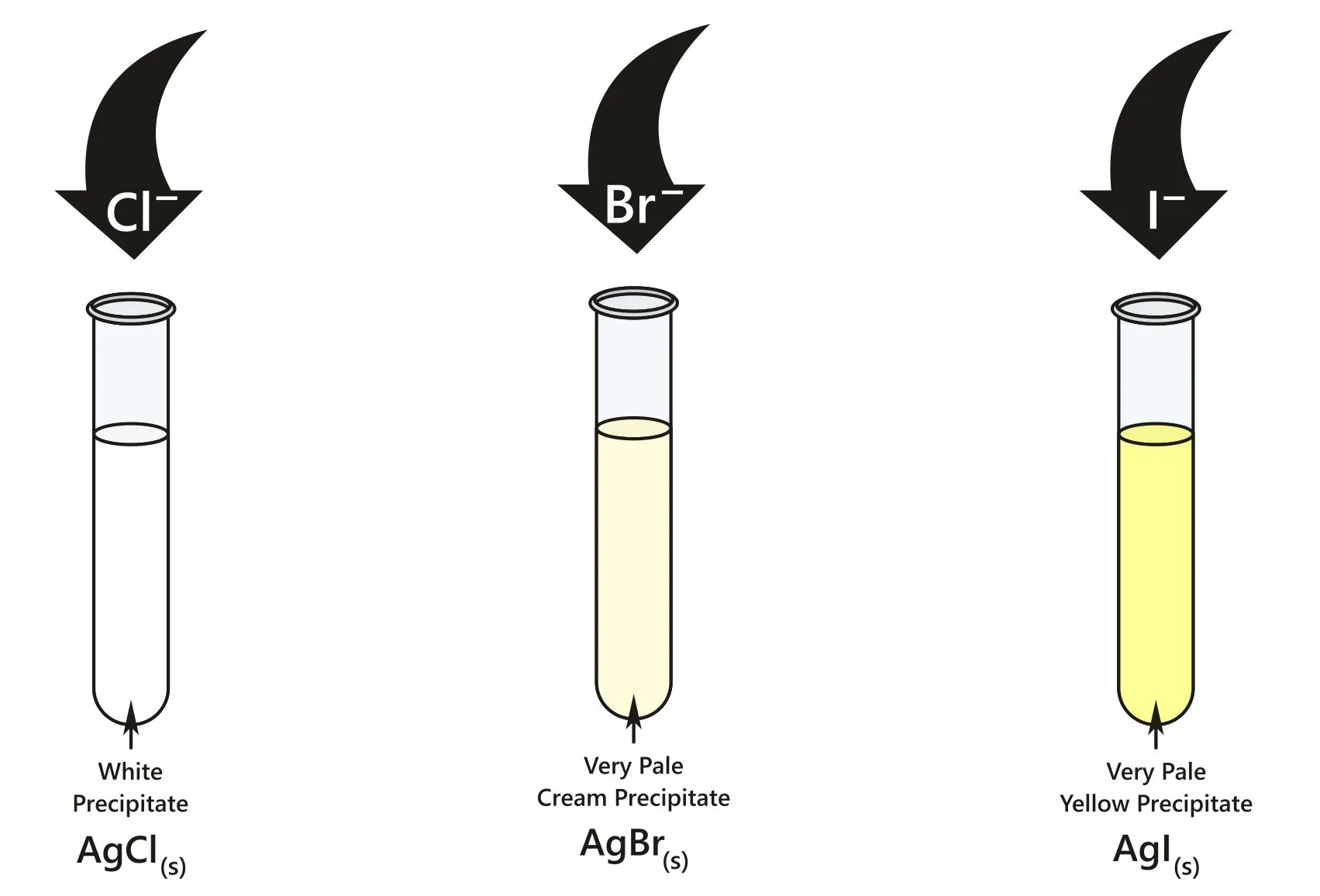
- \( \mathrm{AgCl} \): white precipitate (slow formation)
- \( \mathrm{AgBr} \): cream precipitate (moderate rate)
- \( \mathrm{AgI} \): yellow precipitate (fast formation)
Interpretation of Results
- A faster formation of precipitate indicates a faster rate of hydrolysis.
- Tertiary halogenoalkanes react fastest due to stable carbocation formation (\( \mathrm{S_N1} \)).
- Primary halogenoalkanes react slowest as they undergo \( \mathrm{S_N2} \).
- Iodoalkanes react faster than bromoalkanes and chloroalkanes due to weaker \( \mathrm{C-X} \) bonds.
Sources of Error
- Difficulty in judging the exact time when the precipitate first appears.
- Inconsistent temperature affecting reaction rates.
- Inaccurate measurement of volumes.
Improvements
- Use a colorimeter to measure turbidity for more accurate detection of precipitate formation.
- Use a thermostatically controlled water bath to maintain constant temperature.
- Repeat experiments and calculate average values to improve reliability.
Example 1:
Explain why ethanol is used in this experiment.
▶️ Answer/Explanation
Halogenoalkanes are not soluble in water but are soluble in organic solvents such as ethanol.
Ethanol acts as a solvent to dissolve the halogenoalkane and allow it to mix with the aqueous silver nitrate solution.
This ensures that the reaction can occur effectively throughout the mixture.
Example 2:
A student observes that one halogenoalkane forms a precipitate almost immediately, while another takes several minutes. Explain this observation.
▶️ Answer/Explanation
The halogenoalkane that forms a precipitate quickly has a faster rate of hydrolysis.
This may be due to a weaker \( \mathrm{C-X} \) bond (e.g. \( \mathrm{C-I} \)) or a more stable carbocation (tertiary structure).
The slower reaction may involve a stronger bond (e.g. \( \mathrm{C-Cl} \)) or a primary structure that reacts via the slower \( \mathrm{S_N2} \) mechanism.
Therefore, differences in bond strength and mechanism explain the variation in reaction rates.
