Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.12 Reactivity trends-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.12 Reactivity trends- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.12 Reactivity trends- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.12 know the trend in reactivity of primary, secondary and tertiary halogenoalkanes
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.12 Trend in Reactivity of Primary, Secondary and Tertiary Halogenoalkanes
The reactivity of halogenoalkanes in nucleophilic substitution reactions depends on their structure, specifically whether they are primary, secondary or tertiary. This affects both the mechanism of reaction and the stability of intermediates formed.
Overall Trend in Reactivity

Tertiary > Secondary > Primary
Explanation of the Trend
- Tertiary halogenoalkanes react fastest because they readily form stable carbocations.
- Secondary halogenoalkanes have moderate reactivity due to intermediate carbocation stability.
- Primary halogenoalkanes react slowest as they do not form stable carbocations.
Link to Reaction Mechanisms
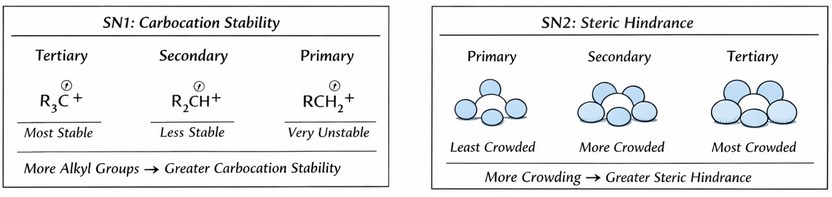
- Tertiary halogenoalkanes react mainly via the \( \mathrm{S_N1} \) mechanism.
- This involves formation of a carbocation intermediate.
- Carbocation stability increases with more alkyl groups due to electron-donating effects.
- Primary halogenoalkanes react via the \( \mathrm{S_N2} \) mechanism.
- This requires direct collision with the nucleophile.
- Although primary halogenoalkanes have low steric hindrance, the absence of carbocation formation makes the overall process slower compared to \( \mathrm{S_N1} \).
Role of Carbocation Stability
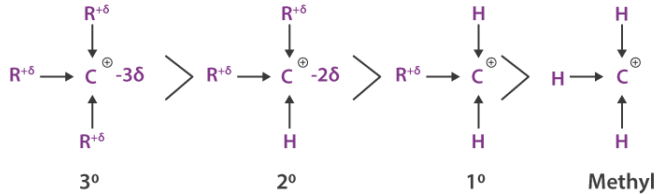
- Tertiary carbocations are stabilised by three alkyl groups.
- Alkyl groups donate electron density, reducing the positive charge on the carbon.
- This lowers the energy required for bond breaking and increases reaction rate.
Summary
- Reactivity increases with increasing substitution of the carbon attached to the halogen.
- Mechanism changes from \( \mathrm{S_N2} \) (primary) to \( \mathrm{S_N1} \) (tertiary).
- Stability of intermediates is the key factor controlling reactivity.
Example 1:
Explain why tertiary halogenoalkanes are more reactive than primary halogenoalkanes in hydrolysis reactions.
▶️ Answer/Explanation
Tertiary halogenoalkanes react via the \( \mathrm{S_N1} \) mechanism, which involves formation of a carbocation intermediate.
The tertiary carbocation is stabilised by three alkyl groups through electron-donating effects.
This stabilisation lowers the energy required for bond breaking, increasing the rate of reaction.
Primary halogenoalkanes do not form stable carbocations and react via the slower \( \mathrm{S_N2} \) mechanism.
Example 2:
A student compares the rates of reaction of 1-bromopropane, 2-bromopropane and 2-bromo-2-methylpropane. Predict and explain the order of reactivity.
▶️ Answer/Explanation
2-bromo-2-methylpropane (tertiary) reacts fastest, followed by 2-bromopropane (secondary), then 1-bromopropane (primary).
The tertiary halogenoalkane forms the most stable carbocation due to three alkyl groups.
The secondary carbocation is less stable, and the primary carbocation is highly unstable.
Therefore, the rate of reaction follows the trend tertiary > secondary > primary.
