Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.13 Bond enthalpy and reactivity-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.13 Bond enthalpy and reactivity- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.13 Bond enthalpy and reactivity- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.13 understand, in terms of bond enthalpy, the trend in reactivity of chloro-, bromo- and iodoalkanes
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.13 Reactivity of Chloro-, Bromo- and Iodoalkanes in Terms of Bond Enthalpy
The reactivity of halogenoalkanes in nucleophilic substitution reactions depends on how easily the carbon–halogen bond can be broken. This is determined by the bond enthalpy of the \( \mathrm{C-X} \) bond, which is the energy required to break the bond.
Trend in Reactivity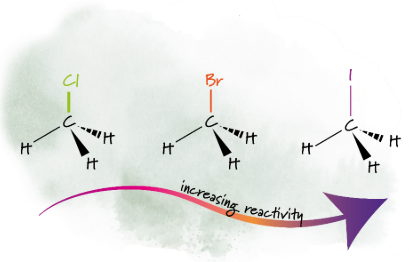
Iodoalkanes > Bromoalkanes > Chloroalkanes
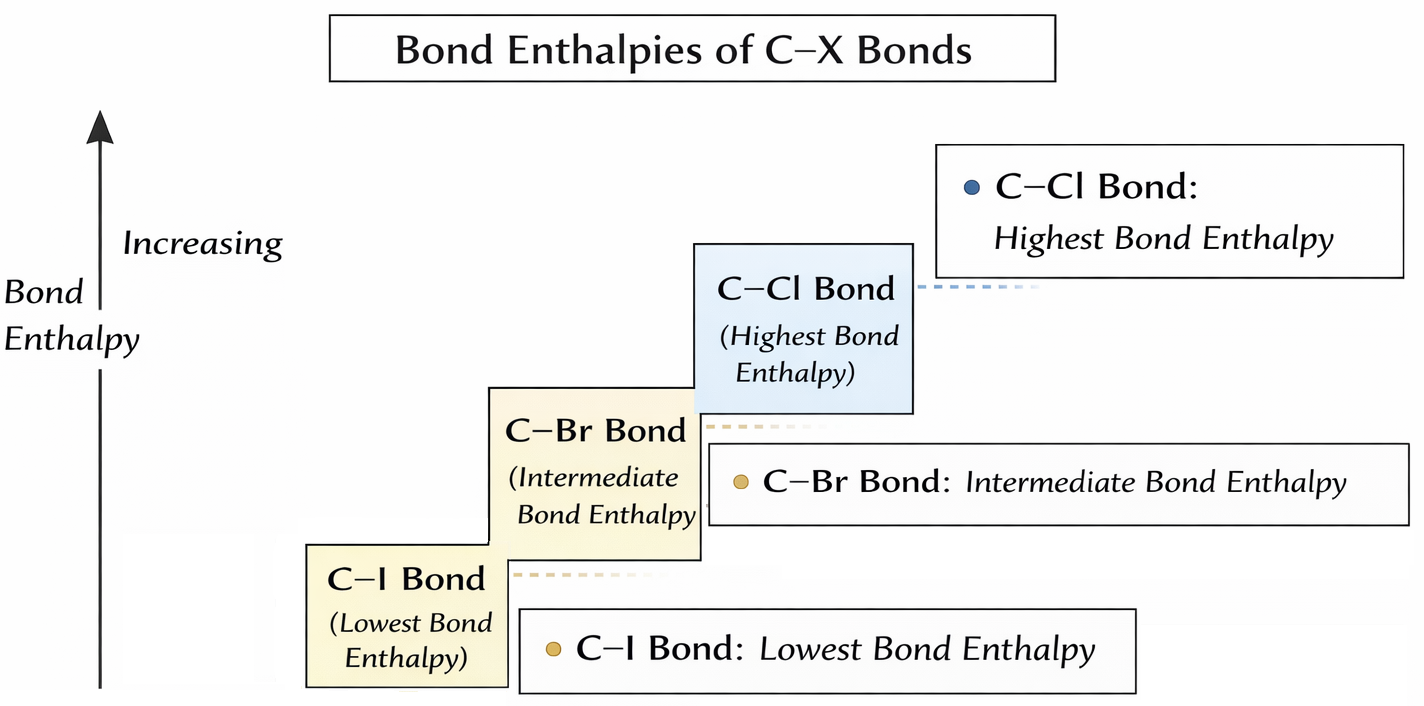
Bond Enthalpy and Bond Strength
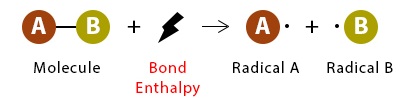
Bond enthalpy is the energy required to break one mole of bonds in the gaseous state. Lower bond enthalpy means a weaker bond that is easier to break.
- The \( \mathrm{C-I} \) bond has the lowest bond enthalpy.
- The \( \mathrm{C-Br} \) bond has intermediate bond enthalpy.

- The \( \mathrm{C-Cl} \) bond has the highest bond enthalpy.
Explanation of the Trend
- As the halogen atom size increases from Cl to Br to I, the bond length increases.
- Larger atoms have poorer orbital overlap with carbon, resulting in weaker bonds.
- Therefore, less energy is required to break the \( \mathrm{C-I} \) bond compared to \( \mathrm{C-Cl} \).
Link to Reactivity
- Reactions involving halogenoalkanes require breaking the \( \mathrm{C-X} \) bond.
- Weaker bonds break more easily, leading to faster reactions.
- Stronger bonds require more energy to break, resulting in slower reactions.
Therefore, iodoalkanes are the most reactive and chloroalkanes are the least reactive in nucleophilic substitution reactions.
Key Points
- Reactivity is inversely related to bond enthalpy.
- Larger halogens form longer, weaker bonds.
- Bond breaking is the rate-determining step in many substitution reactions.
Example 1:
Explain why iodoalkanes are more reactive than chloroalkanes in nucleophilic substitution reactions.
▶️ Answer/Explanation
The \( \mathrm{C-I} \) bond has a lower bond enthalpy than the \( \mathrm{C-Cl} \) bond.
This means less energy is required to break the bond in iodoalkanes.
The larger iodine atom results in poorer orbital overlap, weakening the bond further.
Therefore, iodoalkanes react faster because the bond to be broken is weaker.
Example 2:
A student observes that a bromoalkane reacts faster than a chloroalkane but slower than an iodoalkane. Explain this trend using bond enthalpy.
▶️ Answer/Explanation
The \( \mathrm{C-Br} \) bond has a bond enthalpy intermediate between \( \mathrm{C-Cl} \) and \( \mathrm{C-I} \).
It is weaker than the \( \mathrm{C-Cl} \) bond, so it breaks more easily, leading to faster reaction than chloroalkanes.
However, it is stronger than the \( \mathrm{C-I} \) bond, so more energy is required to break it compared to iodoalkanes.
Therefore, the reactivity follows the order iodoalkane > bromoalkane > chloroalkane.