Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.14 CORE PRACTICAL 6: Chlorination-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.14 CORE PRACTICAL 6: Chlorination- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.14 CORE PRACTICAL 6: Chlorination- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.14 CORE PRACTICAL 6
Chlorination of 2-methylpropan-2-ol with concentrated hydrochloric acid
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.14 Core Practical 6: Chlorination of 2-methylpropan-2-ol with Concentrated Hydrochloric Acid
This practical investigates the reaction of a tertiary alcohol, 2-methylpropan-2-ol, with concentrated hydrochloric acid to form a tertiary halogenoalkane.
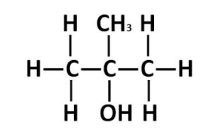
The reaction proceeds rapidly via a nucleophilic substitution mechanism and demonstrates the high reactivity of tertiary alcohols.
Principle of the Reaction
2-methylpropan-2-ol reacts with concentrated \( \mathrm{HCl} \) to form 2-chloro-2-methylpropane and water.
\( \mathbf{(CH_3)_3COH + HCl \rightarrow (CH_3)_3CCl + H_2O} \)
The reaction proceeds via an \( \mathrm{S_N1} \) mechanism due to the formation of a stable tertiary carbocation.
Mechanism (SN1)
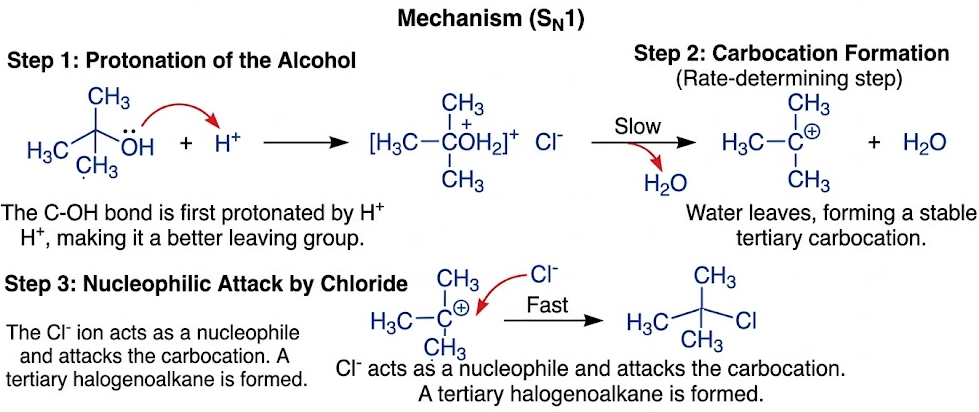
- The \( \mathrm{C-OH} \) bond is first protonated by \( \mathrm{H^+} \), making it a better leaving group.
- Water leaves, forming a stable tertiary carbocation.
- The \( \mathrm{Cl^-} \) ion acts as a nucleophile and attacks the carbocation.
- A tertiary halogenoalkane is formed.
Observations
- The mixture becomes cloudy or forms two layers.
- The halogenoalkane formed is insoluble in water, producing a cloudy appearance.
- The reaction occurs rapidly at room temperature.
Explanation of Observations
- The tertiary carbocation forms easily due to stabilisation by three alkyl groups.
- This leads to a fast \( \mathrm{S_N1} \) reaction.
- The product (2-chloro-2-methylpropane) is non-polar and immiscible with the aqueous layer.
Variables
- Independent variable: type of alcohol (primary, secondary, tertiary).
- Dependent variable: rate of formation of cloudiness or product.
- Controlled variables: temperature, volume of reagents, concentration of acid.
Key Points
- Tertiary alcohols react rapidly with concentrated \( \mathrm{HCl} \).
- The reaction proceeds via a carbocation intermediate.
- This practical demonstrates the effect of structure on reaction rate.
Example 1:
Explain why 2-methylpropan-2-ol reacts rapidly with concentrated hydrochloric acid.
▶️ Answer/Explanation
2-methylpropan-2-ol is a tertiary alcohol, so it forms a stable tertiary carbocation when the \( \mathrm{C-O} \) bond breaks.
The carbocation is stabilised by three alkyl groups through electron-donating effects.
This lowers the energy required for bond breaking, allowing the reaction to proceed rapidly via an \( \mathrm{S_N1} \) mechanism.
Example 2:
Explain why a cloudy layer forms during the reaction.
▶️ Answer/Explanation
The product formed, 2-chloro-2-methylpropane, is a non-polar halogenoalkane.
It is insoluble in the aqueous reaction mixture.
As it forms, it separates from the aqueous layer, causing the mixture to become cloudy or form two layers.
