Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.15 Nomenclature and structures-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.15 Nomenclature and structures- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.15 Nomenclature and structures- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.15 understand the nomenclature of alcohols and be able to draw their structural, displayed and skeletal formulae
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.15 Nomenclature and Representation of Alcohols
Alcohols are organic compounds containing the hydroxyl functional group \( \mathrm{-OH} \) attached to a saturated carbon atom. They are an important homologous series and follow systematic IUPAC naming rules. Alcohols can also be represented using structural, displayed and skeletal formulae.
Nomenclature of Alcohols
Alcohols are named by identifying the longest carbon chain containing the \( \mathrm{-OH} \) group and replacing the ending “-ane” with “-ol”.
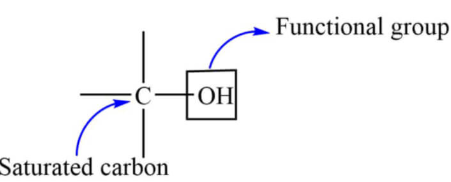
- The longest carbon chain containing the \( \mathrm{-OH} \) group is selected as the parent chain.
- The chain is numbered so that the carbon bearing the \( \mathrm{-OH} \) group has the lowest possible number.
- The position of the \( \mathrm{-OH} \) group is indicated using a number.
- The suffix “-ol” is used instead of “-ane”.
- If there are substituents, they are named and numbered as in alkanes.
Examples of Naming
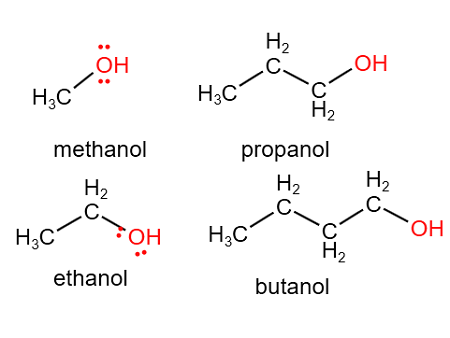
\( \mathrm{CH_3CH_2OH} \) → ethanol
\( \mathrm{CH_3CH(OH)CH_3} \) → propan-2-ol
Structural Formula
A structural formula shows how atoms are arranged and connected, but bonds are written in a condensed form.
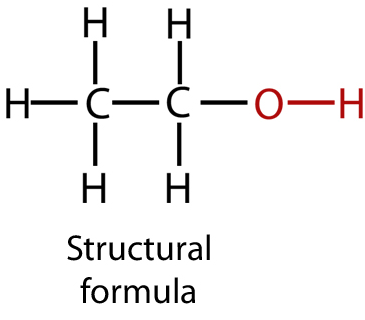
- It shows the grouping of atoms, e.g. \( \mathrm{CH_3CH_2OH} \).
- The \( \mathrm{-OH} \) group is clearly shown at the end or within the chain.
Displayed Formula
A displayed formula shows all atoms and all bonds explicitly.
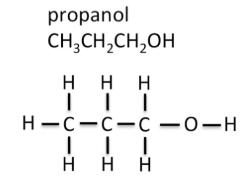
- Every covalent bond is drawn.
- The \( \mathrm{O-H} \) bond is clearly visible.
- Useful for showing bonding and structure in detail.
Skeletal Formula
A skeletal formula represents the carbon framework using lines, with carbon atoms and most hydrogen atoms omitted.
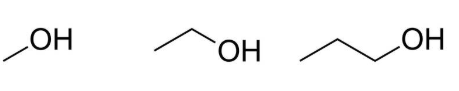
- Each line represents a bond between carbon atoms.
- Carbon atoms are assumed at the ends and intersections of lines.
- Hydrogen atoms attached to carbon are not shown.
- Functional groups such as \( \mathrm{-OH} \) are always shown.
Skeletal formulae provide a simplified way to represent complex organic molecules while retaining essential structural information.
Example 1:
Name the compound with structural formula \( \mathrm{CH_3CH(OH)CH_2CH_3} \) and explain your answer.
▶️ Answer/Explanation
The longest carbon chain contains four carbon atoms, so the parent name is butane.
The \( \mathrm{-OH} \) group is attached to the second carbon when numbered to give the lowest possible number.
Therefore, the compound is named butan-2-ol.
Example 2:
Draw and explain the skeletal formula of propan-1-ol.
▶️ Answer/Explanation
Propan-1-ol has a three-carbon chain with the \( \mathrm{-OH} \) group attached to the first carbon.
In the skeletal formula, this is shown as a two-line zigzag representing three carbons, with the \( \mathrm{-OH} \) group attached to one end.
Carbon and hydrogen atoms are not explicitly shown, but the structure implies their presence.
