Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.16 Types of alcohols-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.16 Types of alcohols- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.16 Types of alcohols- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.16 understand the distinction between primary, secondary and tertiary alcohols
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.16 Primary, Secondary and Tertiary Alcohols
Alcohols can be classified as primary, secondary or tertiary depending on the number of alkyl groups attached to the carbon atom that is bonded to the hydroxyl group \( \mathrm{-OH} \). This classification is important because it affects the chemical properties and reactivity of the alcohol.
Classification of Alcohols
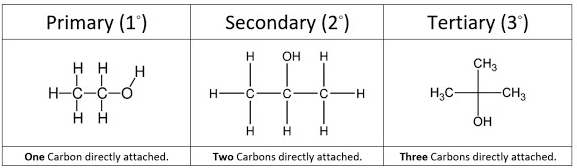
The type of alcohol depends on how many carbon atoms are directly bonded to the carbon bearing the \( \mathrm{-OH} \) group.
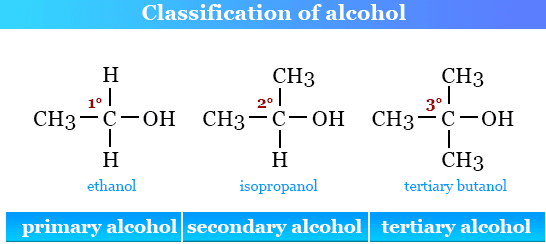
- Primary (1°) alcohol: the carbon bonded to the \( \mathrm{-OH} \) group is attached to only one other carbon atom.
- Secondary (2°) alcohol: the carbon bonded to the \( \mathrm{-OH} \) group is attached to two other carbon atoms.
- Tertiary (3°) alcohol: the carbon bonded to the \( \mathrm{-OH} \) group is attached to three other carbon atoms.
Structural Representation
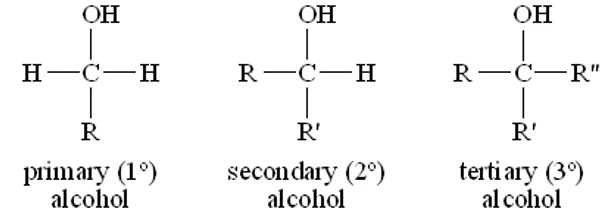
- Primary: \( \mathrm{RCH_2OH} \)
- Secondary: \( \mathrm{R_2CHOH} \)
- Tertiary: \( \mathrm{R_3COH} \)
Explanation of the Distinction
- In a primary alcohol, the \( \mathrm{-OH} \)-bearing carbon is at the end of the chain or attached to only one alkyl group.
- In a secondary alcohol, the \( \mathrm{-OH} \)-bearing carbon is located within the chain and bonded to two alkyl groups.
- In a tertiary alcohol, the \( \mathrm{-OH} \)-bearing carbon is bonded to three alkyl groups, making it highly substituted.
The degree of substitution affects steric hindrance and the stability of intermediates formed during reactions, which leads to differences in reactivity between primary, secondary and tertiary alcohols.
Examples
- Primary: \( \mathrm{CH_3CH_2OH} \) (ethanol)
- Secondary: \( \mathrm{CH_3CH(OH)CH_3} \) (propan-2-ol)
- Tertiary: \( \mathrm{(CH_3)_3COH} \) (2-methylpropan-2-ol)
Key Features of the Distinction
- The classification depends only on the number of carbon atoms attached to the \( \mathrm{-OH} \)-bearing carbon.
- It is independent of the total number of carbon atoms in the molecule.
- This distinction is important for predicting chemical behaviour, such as oxidation and substitution reactions.
Example 1:
Classify the alcohol \( \mathrm{CH_3CH(OH)CH_2CH_3} \) and explain your answer.
▶️ Answer/Explanation
The carbon bearing the \( \mathrm{-OH} \) group is bonded to two other carbon atoms.
Therefore, it is a secondary alcohol.
This is because the classification depends on the number of alkyl groups attached to the \( \mathrm{-OH} \)-bearing carbon.
Example 2:
Explain why \( \mathrm{(CH_3)_3COH} \) is classified as a tertiary alcohol.
▶️ Answer/Explanation
In \( \mathrm{(CH_3)_3COH} \), the carbon atom bonded to the \( \mathrm{-OH} \) group is attached to three other carbon atoms.
This means it has three alkyl groups attached.
Therefore, it is classified as a tertiary alcohol.
The high degree of substitution also affects its chemical reactivity compared to primary and secondary alcohols.
