Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.17 Reactions of alcohols-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – Link- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – Link- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.17 understand the reactions of alcohols with:
i oxygen in air (combustion)
ii halogenating agents
• PCl₅ to produce chloroalkanes (including its use as a qualitative test for the –OH group)
• 50% concentrated sulfuric acid and potassium bromide to produce bromoalkanes
• red phosphorus and iodine to produce iodoalkanes
iii concentrated phosphoric acid to form alkenes by elimination
Descriptions of the mechanisms of these reactions are not required
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.17 Reactions of Alcohols
Alcohols undergo a range of important reactions due to the presence of the hydroxyl group \( \mathrm{-OH} \). These include combustion, substitution with halogenating agents, and elimination to form alkenes. The reactions involve breaking the \( \mathrm{C-O} \) or \( \mathrm{O-H} \) bonds, and the behaviour of alcohols can be explained by the polarity of the \( \mathrm{O-H} \) bond and the ability of the \( \mathrm{-OH} \) group to be replaced or removed.
(i) Combustion of Alcohols
Alcohols undergo complete combustion in oxygen to produce carbon dioxide and water.
\( \mathrm{C_2H_5OH + 3O_2 \rightarrow 2CO_2 + 3H_2O} \)
- Combustion is an exothermic reaction, releasing a large amount of energy.
- Alcohols burn with a clean flame due to their oxygen content.
- They can be used as fuels or fuel additives.
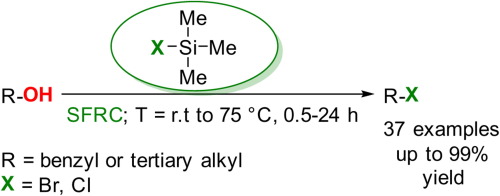
(ii) Reaction with Halogenating Agents
Alcohols can be converted into haloalkanes by replacing the \( \mathrm{-OH} \) group with a halogen atom.
Reaction with \( \mathrm{PCl_5} \)
\( \mathrm{ROH + PCl_5 \rightarrow RCl + POCl_3 + HCl} \)
- The \( \mathrm{-OH} \) group is replaced by chlorine to form a chloroalkane.
- Hydrogen chloride gas is produced, observed as steamy white fumes.
- This reaction is used as a qualitative test for the presence of the \( \mathrm{-OH} \) group.
Reaction with \( \mathrm{KBr} \) and concentrated \( \mathrm{H_2SO_4} \)
\( \mathrm{ROH + HBr \rightarrow RBr + H_2O} \)
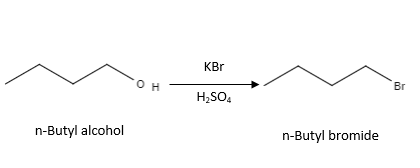
Hydrogen bromide is generated in situ from potassium bromide and concentrated sulfuric acid:
\( \mathrm{KBr + H_2SO_4 \rightarrow HBr + KHSO_4} \)
- The \( \mathrm{-OH} \) group is replaced by bromine to form a bromoalkane.
- This reaction proceeds via substitution.
Reaction with red phosphorus and iodine
\( \mathrm{ROH + HI \rightarrow RI + H_2O} \)
Hydrogen iodide is formed :
\( \mathrm{P + I_2 + H_2O \rightarrow HI} \)
- The \( \mathrm{-OH} \) group is replaced by iodine to form an iodoalkane.
- This method is necessary because hydrogen iodide is unstable and must be generated during the reaction.
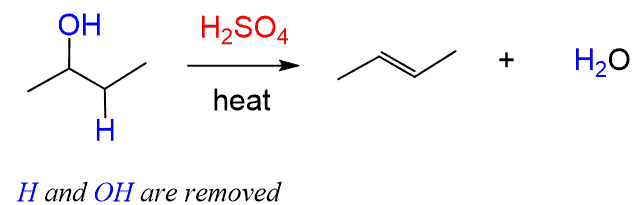
(iii) Elimination to form Alkenes
Alcohols can undergo elimination when heated with concentrated phosphoric acid to form alkenes.
\( \mathrm{C_2H_5OH \rightarrow C_2H_4 + H_2O} \)
- The reaction involves removal of a molecule of water (dehydration).
- A double bond forms between carbon atoms, producing an alkene.
- Concentrated phosphoric acid acts as a catalyst and dehydrating agent.
Key Features of Alcohol Reactions
- Alcohols can undergo combustion, substitution and elimination reactions.
- The \( \mathrm{-OH} \) group can be replaced by halogens or removed as water.
- Reaction conditions determine the type of reaction that occurs.
- No mechanism detail is required, only overall reaction understanding.
Example 1:
Describe the observations and products when ethanol reacts with \( \mathrm{PCl_5} \).
▶️ Answer/Explanation
Ethanol reacts with \( \mathrm{PCl_5} \) to form chloroethane, phosphorus oxychloride and hydrogen chloride.
\( \mathrm{C_2H_5OH + PCl_5 \rightarrow C_2H_5Cl + POCl_3 + HCl} \)
Steamy white fumes of hydrogen chloride gas are observed.
This confirms the presence of the \( \mathrm{-OH} \) group in the alcohol.
Example 2:
Explain how propan-2-ol can be converted into propene and state the conditions required.
▶️ Answer/Explanation
Propan-2-ol is heated with concentrated phosphoric acid, which acts as a dehydrating agent.
A molecule of water is eliminated, forming propene.
\( \mathrm{CH_3CH(OH)CH_3 \rightarrow CH_3CH=CH_2 + H_2O} \)
This is an elimination reaction in which a double bond is formed between carbon atoms.
