Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.18 Oxidation of alcohols-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.18 Oxidation of alcohols- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.18 Oxidation of alcohols- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.18 understand that potassium dichromate(VI) in dilute sulfuric acid can oxidise:
i primary alcohols to produce aldehydes (which give a positive result with Benedict’s or Fehling’s solution) if the product is distilled as it forms
ii primary alcohols to produce carboxylic acids (which give a positive result with sodium carbonate or sodium hydrogencarbonate) if the reagents are heated under reflux
iii secondary alcohols to produce ketones
In equations, the oxidising agent can be represented by [O]
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
F10.18 Oxidation of Alcohols using Potassium Dichromate(VI)
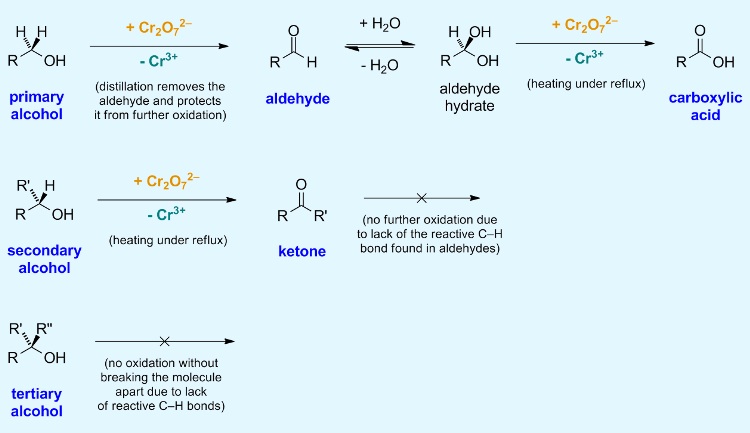
Potassium dichromate(VI), \( \mathrm{K_2Cr_2O_7} \), in dilute sulfuric acid is a strong oxidising agent used to oxidise alcohols. The extent of oxidation depends on the type of alcohol and the reaction conditions. Primary alcohols can be oxidised to aldehydes or further to carboxylic acids, while secondary alcohols are oxidised to ketones. Tertiary alcohols are not oxidised under these conditions.
Oxidising Agent
The oxidising agent can be represented as \( \mathrm{[O]} \) in equations.
During oxidation, the orange dichromate(VI) ions are reduced to green chromium(III) ions, providing a visible colour change.
(i) Oxidation of Primary Alcohols to Aldehydes
\( \mathrm{RCH_2OH + [O] \rightarrow RCHO + H_2O} \)
- Primary alcohols are first oxidised to aldehydes.
- The aldehyde must be distilled off as it forms to prevent further oxidation.
- Aldehydes can be identified using Benedict’s or Fehling’s solution, giving a positive result.
This occurs because aldehydes are easily oxidised further, so removing them prevents further reaction.
(ii) Oxidation of Primary Alcohols to Carboxylic Acids
\( \mathrm{RCH_2OH + 2[O] \rightarrow RCOOH + H_2O} \)
- If the reaction mixture is heated under reflux, the aldehyde formed is not removed.
- It is further oxidised to a carboxylic acid.
- Carboxylic acids react with sodium carbonate or sodium hydrogencarbonate to produce carbon dioxide gas (effervescence).
Reflux allows the reaction to proceed to completion by preventing loss of volatile intermediates.
(iii) Oxidation of Secondary Alcohols to Ketones
\( \mathrm{R_2CHOH + [O] \rightarrow R_2CO + H_2O} \)
- Secondary alcohols are oxidised to ketones.
- Ketones are not easily oxidised further under these conditions.
- Therefore, oxidation stops at the ketone stage.
Summary of Conditions
- Distillation: used to obtain aldehydes from primary alcohols.
- Reflux: used to obtain carboxylic acids from primary alcohols.
- Acidified \( \mathrm{K_2Cr_2O_7} \): oxidising agent in all cases.
Key Features of Alcohol Oxidation
- The type of alcohol determines the oxidation product.
- Reaction conditions control the extent of oxidation.
- A colour change from orange to green indicates oxidation has occurred.
- Tertiary alcohols are not oxidised under these conditions due to the absence of a hydrogen on the \( \mathrm{-OH} \)-bearing carbon.
Example 1:
Explain how ethanol can be oxidised to ethanal and why distillation is required.
▶️ Answer/Explanation
Ethanol is a primary alcohol and can be oxidised using acidified potassium dichromate(VI).
\( \mathrm{CH_3CH_2OH + [O] \rightarrow CH_3CHO + H_2O} \)
The aldehyde formed (ethanal) is easily oxidised further to a carboxylic acid.
Therefore, distillation is used to remove ethanal as soon as it forms, preventing further oxidation.
Example 2:
A compound is oxidised using acidified potassium dichromate(VI) to form a ketone. Deduce the type of alcohol and explain your answer.
▶️ Answer/Explanation
The formation of a ketone indicates that the original alcohol was a secondary alcohol.
Secondary alcohols are oxidised to ketones when treated with acidified potassium dichromate(VI).
Primary alcohols would form aldehydes or carboxylic acids, while tertiary alcohols are not oxidised.
Therefore, the alcohol must be secondary.
