Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.19 Preparation and purification-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.19 Preparation and purification- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.19 Preparation and purification- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.19 understand the following techniques in the preparation and purification of a liquid organic compound:
i heating under reflux
ii extraction with a solvent using a separating funnel
iii distillation
iv drying with an anhydrous salt
v boiling temperature determination
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.19 Preparation and Purification Techniques for Liquid Organic Compounds
A range of experimental techniques are used in organic chemistry to prepare and purify liquid compounds. These methods ensure reactions proceed efficiently and that the final product is obtained in a pure, dry form. Each technique is based on a specific physical property such as boiling temperature, solubility or affinity for water.
(i) Heating under Reflux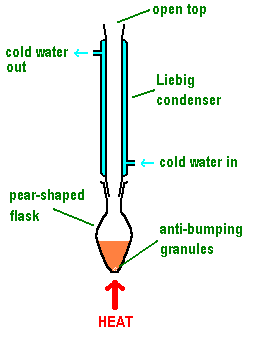
Heating under reflux allows a reaction mixture to be heated continuously at its boiling temperature without loss of volatile substances.
- The apparatus consists of a round-bottom flask connected to a vertical condenser.
- Cold water flows through the condenser (in at the bottom, out at the top) to ensure efficient cooling.
- Vapours formed during heating rise into the condenser, cool, and condense back into liquid.
- The condensed liquid returns to the reaction flask, preventing loss of reactants or products.
- Anti-bumping granules are added to ensure smooth boiling and prevent sudden violent boiling.
Reflux is essential when reactions are slow and require prolonged heating to reach completion, particularly in oxidation or substitution reactions.
(ii) Extraction using a Separating Funnel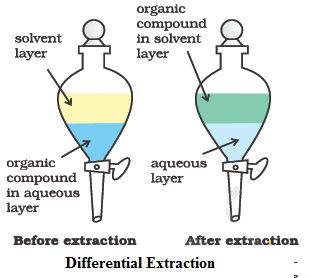
Extraction separates compounds based on their different solubilities in two immiscible liquids, typically an aqueous layer and an organic solvent.
- The mixture is placed in a separating funnel and an organic solvent is added.
- The funnel is shaken and inverted, with the tap opened periodically to release pressure.
- Two distinct layers form due to immiscibility of the liquids.
- The distribution of the compound depends on its relative solubility (partitioning) between the two layers.
- The denser layer is drained first, followed by the less dense layer.
- Multiple extractions with small volumes of solvent are more effective than a single extraction.
This technique is used to isolate organic products from aqueous mixtures and to remove impurities.
(iii) Distillation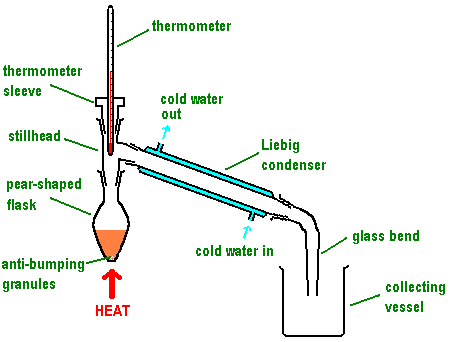
Distillation separates liquids based on differences in boiling temperature and volatility.
- The mixture is heated in a distillation flask fitted with a thermometer and condenser.
- The thermometer bulb is positioned at the entrance to the condenser to measure vapour temperature accurately.
- The component with the lowest boiling temperature vaporises first.
- Vapour passes into the condenser, where it cools and condenses into a liquid.
- The condensed liquid is collected as the distillate.
- Anti-bumping granules are used to ensure smooth boiling.
Simple distillation is suitable when the difference in boiling temperatures is large, while fractional distillation is used when boiling temperatures are closer.
(iv) Drying with an Anhydrous Salt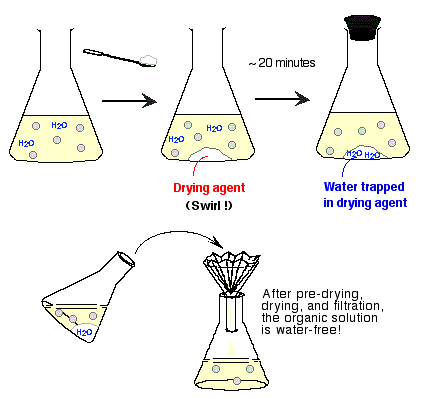
Drying removes residual water from an organic liquid using a drying agent.
- A suitable anhydrous salt (e.g. calcium chloride, magnesium sulfate, sodium sulfate) is added to the organic liquid.
- The drying agent absorbs water and becomes hydrated.
- The mixture is swirled and allowed to stand until the liquid becomes clear.
- Excess drying agent indicates that all water has been removed.
- The drying agent is removed by filtration or decanting.
The choice of drying agent depends on the compound being dried, as some may react with certain functional groups.
(v) Boiling Temperature Determination
The boiling temperature of a liquid is measured to assess its purity and confirm its identity.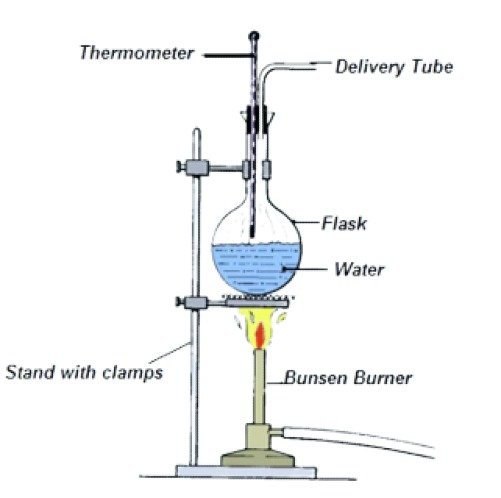
- A pure liquid has a sharp, constant boiling temperature at a given pressure.
- Impurities cause the boiling temperature to be lower or higher and occur over a range.
- The boiling temperature is measured during distillation using a thermometer.
- The recorded value is compared with known data to identify the compound.
- Atmospheric pressure must be considered, as boiling temperature varies with pressure.
A narrow boiling temperature range indicates high purity, while a broad range indicates the presence of impurities.
Key Summary
- Reflux enables prolonged heating without loss of volatile substances.
- Extraction separates substances based on solubility differences.
- Distillation separates liquids based on boiling temperature differences.
- Drying removes water using anhydrous salts.
- Boiling temperature determination assesses purity and identity.
Example 1:
Explain why heating under reflux is preferred over simple heating when preparing an organic liquid.
▶️ Answer/Explanation
Heating under reflux allows the reaction mixture to be heated for a long period without loss of volatile substances.
Vapours are condensed and returned to the flask, ensuring reactants remain in the system.
This increases the extent of reaction and improves yield compared to simple heating where substances may evaporate and be lost.
Example 2:
A liquid has a boiling temperature range rather than a sharp value. Explain what this indicates and how the liquid can be purified.
▶️ Answer/Explanation
A boiling temperature range indicates that the liquid is impure.
Impurities disrupt intermolecular forces, causing boiling to occur over a range of temperatures.
The liquid can be purified by distillation, where the component with the lowest boiling temperature is separated and collected.
