Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.2 Reaction mechanisms-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.2 Reaction mechanisms- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.2 Reaction mechanisms- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.2 understand the concept of a reaction mechanism
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.2 Reaction Mechanisms
A reaction mechanism describes the detailed step-by-step pathway by which a chemical reaction occurs. It shows how bonds are broken and formed, the movement of electrons, and the formation of any intermediate species during the reaction. Understanding mechanisms allows chemists to explain how reactions occur rather than just what the overall change is.
Reaction Mechanism
A reaction mechanism is the sequence of elementary steps that shows how reactants are converted into products, including the movement of electron pairs.
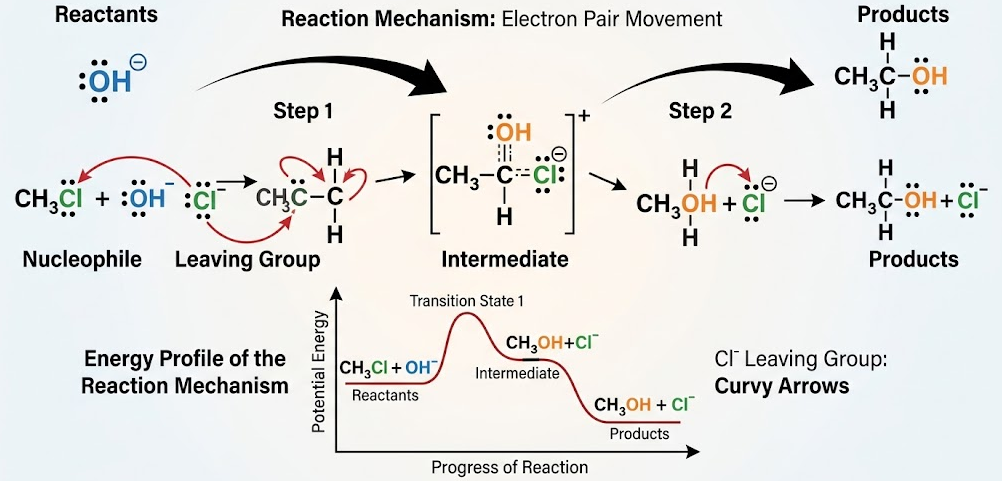
Key Features of Reaction Mechanisms
- Mechanisms show curly arrows to represent the movement of electron pairs.
- Arrows always start from a region of high electron density (such as a lone pair or bond).
- Arrows point towards electron-deficient atoms or regions.
- Bonds are formed when electrons move to form new shared pairs.
- Bonds are broken when electron pairs move away from a bond.
Types of Bond Breaking
Bonds can break in two different ways depending on how electrons are distributed.

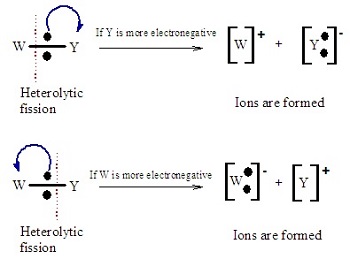
- Homolytic fission: the bond breaks evenly and each atom takes one electron, forming two radicals.
- Heterolytic fission: the bond breaks unevenly and one atom takes both electrons, forming ions.
Reactive Intermediates
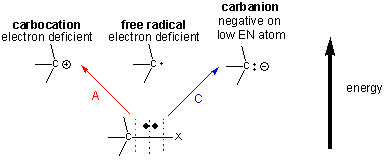
- Free radicals: species with an unpaired electron, formed by homolytic fission.
- Carbocations: positively charged carbon species with an incomplete octet.
- Carbanions: negatively charged carbon species with a lone pair of electrons.
Importance of Mechanisms
- Explain how and why reactions occur.
- Help predict products and reaction pathways.
- Allow understanding of reaction rates and conditions required.
- Provide insight into the role of intermediates and catalysts.
Example 1:
Explain what is meant by a reaction mechanism and why it is important.
▶️ Answer/Explanation
A reaction mechanism is the step-by-step sequence showing how reactants are converted into products, including how bonds are broken and formed.
It involves the movement of electron pairs, often represented using curly arrows.
Understanding mechanisms is important because it explains how reactions occur and allows prediction of products and reaction pathways.
Example 2:
Explain the difference between homolytic and heterolytic bond fission.
▶️ Answer/Explanation
In homolytic fission, the bond breaks evenly and each atom takes one electron from the bond, forming two radicals.
In heterolytic fission, the bond breaks unevenly and one atom takes both electrons, forming a positive ion and a negative ion.
The difference lies in how the bonding electrons are distributed during bond breaking.
