Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.20 CORE PRACTICAL 7: Oxidation of propan-1-ol-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.20 CORE PRACTICAL 7: Oxidation of propan-1-ol- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.20 CORE PRACTICAL 7: Oxidation of propan-1-ol- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.20 CORE PRACTICAL 7
The oxidation of propan-1-ol to produce propanal and propanoic acid
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.20 Core Practical 7: Oxidation of Propan-1-ol
This core practical involves the controlled oxidation of a primary alcohol, propan-1-ol, using acidified potassium dichromate(VI). By changing the reaction conditions, propan-1-ol can be oxidised either to an aldehyde (propanal) or further to a carboxylic acid (propanoic acid). This demonstrates how reaction conditions determine the extent of oxidation.
Principle of the Reaction
Primary alcohols are oxidised first to aldehydes and then further to carboxylic acids using an oxidising agent such as acidified \( \mathrm{K_2Cr_2O_7} \).
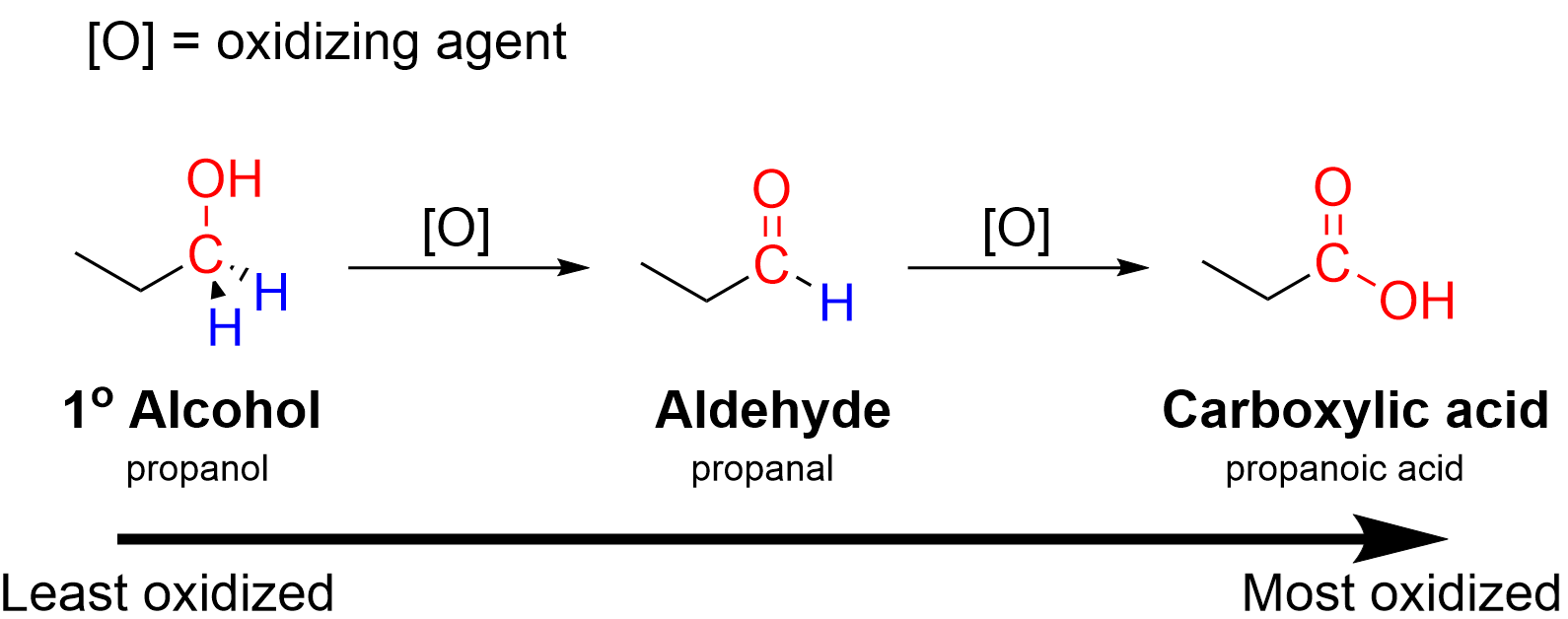
The oxidising agent can be represented as \( \mathrm{[O]} \), and a colour change from orange to green indicates reduction of dichromate(VI) ions to chromium(III) ions.
Oxidation to Propanal (Aldehyde)
\( \mathrm{CH_3CH_2CH_2OH + [O] \rightarrow CH_3CH_2CHO + H_2O} \)
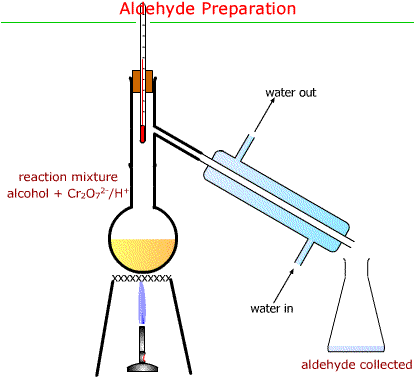
- The reaction mixture is heated gently and distilled.
- Propanal is more volatile than propan-1-ol and is distilled off as it forms.
- Immediate removal prevents further oxidation to the carboxylic acid.
- Anti-bumping granules are used to ensure smooth boiling.
Oxidation to Propanoic Acid
\( \mathrm{CH_3CH_2CH_2OH + 2[O] \rightarrow CH_3CH_2COOH + H_2O} \)
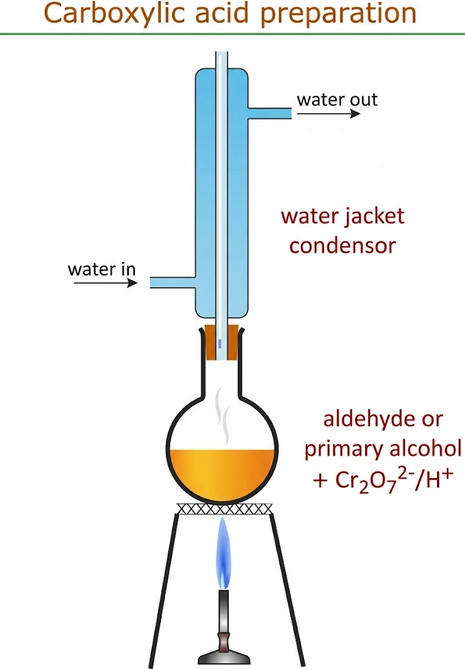
- The reaction mixture is heated under reflux.
- The aldehyde formed is not removed and remains in contact with the oxidising agent.
- Further oxidation occurs to produce propanoic acid.
- Reflux ensures complete oxidation by preventing loss of volatile intermediates.
Experimental Conditions
- Acidified potassium dichromate(VI) is used as the oxidising agent.
- Dilute sulfuric acid provides the acidic conditions required.
- Temperature and apparatus (distillation vs reflux) determine the product formed.
Observations
- The solution changes from orange to green as oxidation occurs.
- A colourless liquid (propanal or propanoic acid) is formed depending on conditions.
- Distillate is collected when forming propanal.
Key Differences in Conditions
- Distillation favours formation of aldehyde by removing it from the reaction mixture.
- Reflux favours formation of carboxylic acid by allowing further oxidation.
- Control of conditions is essential to obtain the desired product.
Testing the Products
- Propanal gives a positive result with Benedict’s or Fehling’s solution (formation of a coloured precipitate).
- Propanoic acid reacts with sodium carbonate or sodium hydrogencarbonate to produce effervescence of \( \mathrm{CO_2} \).
Key Concept
- Primary alcohols can undergo stepwise oxidation.
- Reaction conditions determine whether oxidation stops at the aldehyde or proceeds to the carboxylic acid.
- Careful control of apparatus and temperature is essential in practical work.
Example 1:
A student heats propan-1-ol with acidified \( \mathrm{K_2Cr_2O_7} \) and immediately collects a distillate. Explain why propanal is formed instead of propanoic acid.
▶️ Answer/Explanation
Propan-1-ol is a primary alcohol and is first oxidised to propanal.
The aldehyde formed is more volatile than the alcohol and is distilled off as it forms.
This prevents further oxidation because propanal is removed from the oxidising agent.
Therefore, oxidation stops at the aldehyde stage and propanoic acid is not formed.
Example 2:
Describe how you would modify the conditions to ensure propan-1-ol is fully oxidised to propanoic acid and explain why these conditions are necessary.
▶️ Answer/Explanation
The reaction mixture should be heated under reflux with excess acidified potassium dichromate(VI).
Reflux prevents the escape of the aldehyde intermediate, keeping it in contact with the oxidising agent.
This allows further oxidation of propanal to propanoic acid.
Therefore, complete oxidation occurs due to prolonged heating and continuous contact with the oxidising agent.
