Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.21 Mass spectrometry interpretation-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.21 Mass spectrometry interpretation- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.21 Mass spectrometry interpretation- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.21 be able to interpret data from mass spectra to suggest possible structures of simple organic compounds using the m/z of the molecular ion and fragmentation patterns
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.21 Interpretation of Mass Spectra
Mass spectrometry is an analytical technique used to determine the relative molecular mass and structural features of organic compounds.
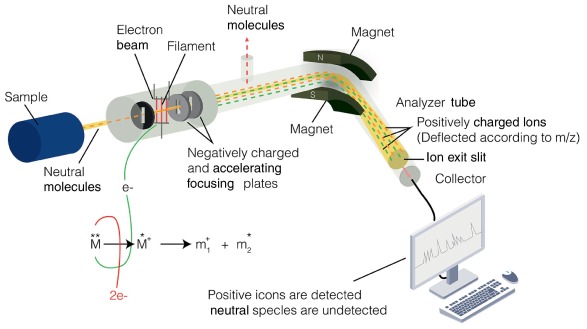
The spectrum produced shows peaks corresponding to ions with different mass-to-charge ratios (\( \mathrm{m/z} \)), which arise from ionisation and fragmentation of molecules. By analysing these peaks, the structure of a compound can be deduced.
Molecular Ion Peak
The molecular ion peak corresponds to the ion formed when a molecule loses one electron without fragmentation.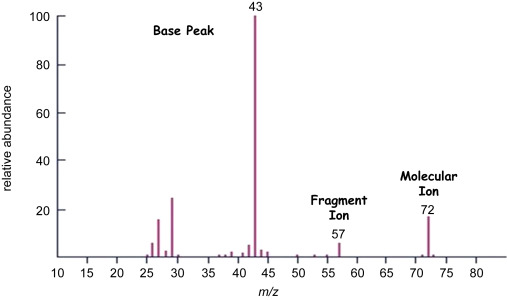
- It is represented as \( \mathrm{M^+} \).
- The \( \mathrm{m/z} \) value of this peak gives the relative molecular mass (\( \mathrm{M_r} \)) of the compound.
- It is usually the peak with the highest \( \mathrm{m/z} \) value in the spectrum.
- The molecular ion peak may be small or absent if the molecule fragments easily.
Base Peak
The base peak is the tallest peak in the spectrum and is assigned 100% relative abundance.
- It corresponds to the most stable and most abundant fragment ion.
- Other peaks are measured relative to the base peak.
Fragmentation Patterns
Fragmentation occurs when the molecular ion breaks into smaller ions and neutral fragments.
![]()
- Each fragment produces a peak at a specific \( \mathrm{m/z} \) value.
- The pattern of peaks provides information about the structure of the molecule.
- More stable carbocations are more likely to form and give more intense peaks.
- Bonds may break in different ways, leading to characteristic fragments.
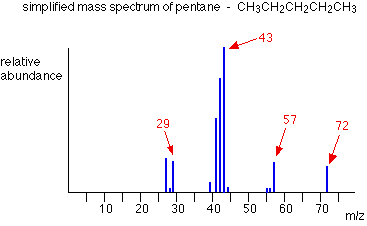
Using \( \mathrm{m/z} \) Values to Deduce Structure
- The molecular ion peak gives the relative molecular mass of the compound.
- Differences between peaks can indicate the loss of specific groups (e.g. \( \mathrm{CH_3} \), \( \mathrm{OH} \)).
- Common fragment ions help identify functional groups and structural features.
- The presence of certain peaks can suggest branching or specific arrangements of atoms.
Typical Fragmentation Features
- Peaks at lower \( \mathrm{m/z} \) values correspond to smaller fragments.
- A peak at \( \mathrm{m/z = 15} \) often indicates a \( \mathrm{CH_3^+} \) fragment.
- Loss of \( \mathrm{H_2O} \) (18 units) is common in alcohols.
- Alkyl groups may break off to form stable carbocations.
Strategy for Interpreting Mass Spectra
- Identify the molecular ion peak to determine \( \mathrm{M_r} \).
- Examine the base peak to identify the most stable fragment.
- Analyse major fragment peaks and their \( \mathrm{m/z} \) values.
- Consider possible bond breakages and functional groups.
- Construct possible structures consistent with all observed data.
Key Features
- The molecular ion peak gives the relative molecular mass.
- Fragmentation patterns provide structural information.
- The base peak represents the most stable fragment ion.
- Interpretation requires combining all spectral data logically.
Example 1:
A compound shows a molecular ion peak at \( \mathrm{m/z = 60} \) and a strong peak at \( \mathrm{m/z = 31} \). Suggest a possible structure and explain your reasoning.
▶️ Answer/Explanation
The molecular ion peak at \( \mathrm{m/z = 60} \) indicates a relative molecular mass of 60.
A common compound with this \( \mathrm{M_r} \) is propan-2-ol (\( \mathrm{C_3H_8O} \)).
The peak at \( \mathrm{m/z = 31} \) corresponds to the \( \mathrm{CH_2OH^+} \) fragment, which is characteristic of alcohols.
Therefore, the compound is likely an alcohol, and a suitable structure is propan-2-ol.
Example 2:
A mass spectrum shows a molecular ion peak at \( \mathrm{m/z = 44} \) and a base peak at \( \mathrm{m/z = 29} \). Suggest a possible structure and explain how the fragmentation supports your answer.
▶️ Answer/Explanation
The molecular ion peak at \( \mathrm{m/z = 44} \) indicates a relative molecular mass of 44.
A possible compound is ethanal (\( \mathrm{CH_3CHO} \)).
The base peak at \( \mathrm{m/z = 29} \) corresponds to a stable \( \mathrm{CHO^+} \) or \( \mathrm{C_2H_5^+} \) fragment.
This fragmentation is consistent with cleavage near the carbonyl group in an aldehyde.
Therefore, the structure is consistent with ethanal.
