Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.22 Infrared spectroscopy-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.22 Infrared spectroscopy- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.22 Infrared spectroscopy- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.22 be able to use infrared spectra, or data from infrared spectra, to deduce functional groups present in organic compounds, and predict infrared absorptions, given wavenumber data, due to familiar functional groups including:
i C–H stretching absorptions in alkanes, alkenes and aldehydes
ii C=C stretching absorption in alkenes
iii O–H stretching absorptions in alcohols and carboxylic acids
iv C=O stretching absorptions in aldehydes, ketones and carboxylic acids
v C–X stretching absorption in halogenoalkanes
vi N–H stretching absorption in amines
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.22 Infrared Spectroscopy
Infrared (IR) spectroscopy is used to identify functional groups in organic compounds by measuring the absorption of infrared radiation.
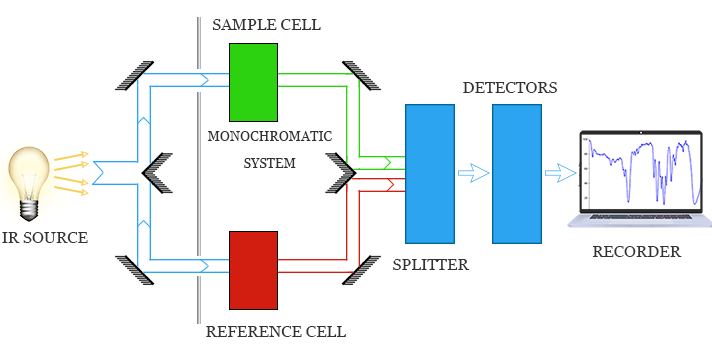
Different bonds absorb IR radiation at characteristic wavenumbers (\( \mathrm{cm^{-1}} \)) due to bond vibrations such as stretching.
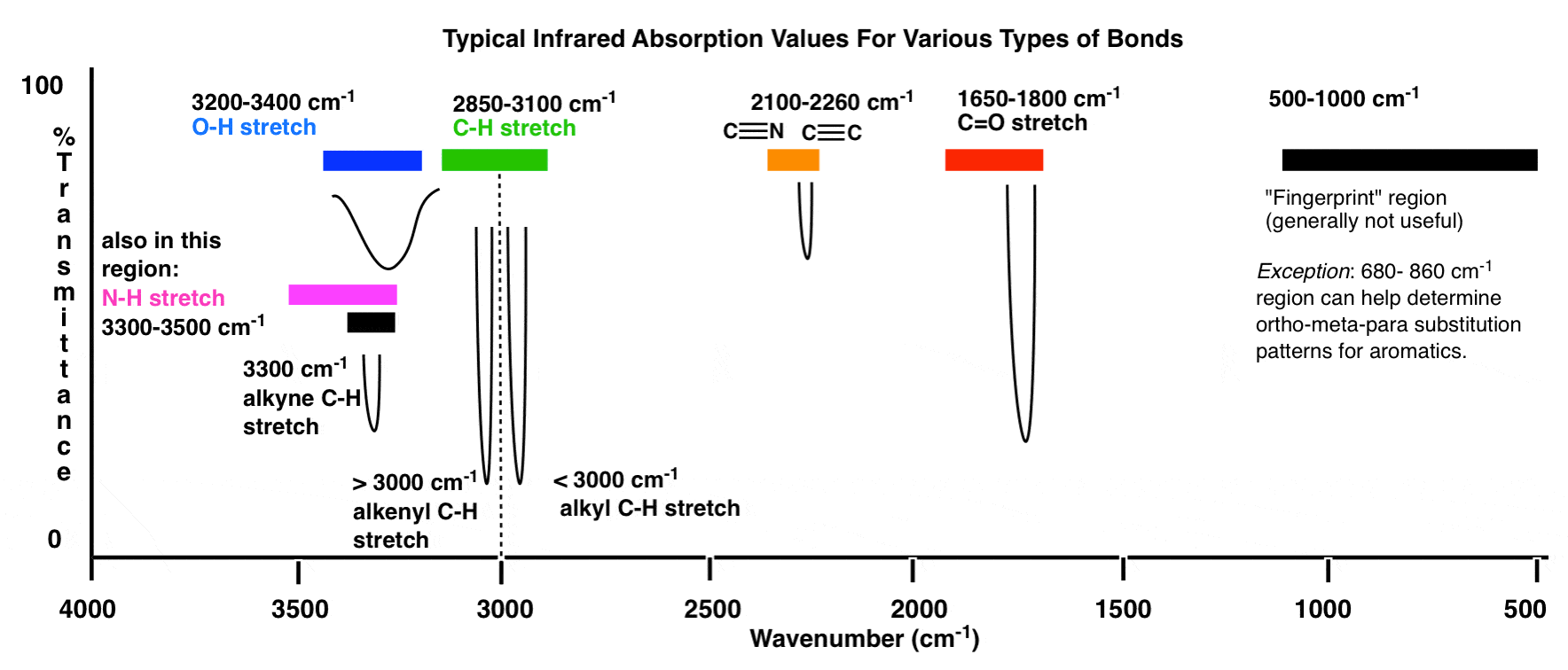
By analysing these absorptions, the presence of specific functional groups can be deduced.
(i) C–H Stretching Absorptions
C–H bonds show characteristic stretching absorptions that vary depending on the type of compound.
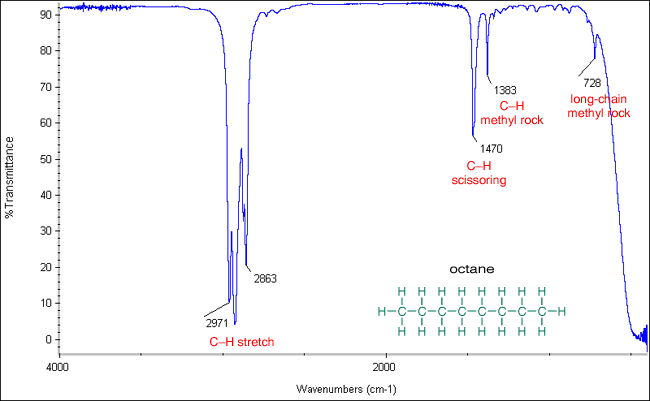
- Alkanes: C–H stretching occurs at approximately \( \mathrm{2850–2960\ cm^{-1}} \).
- Alkenes: C–H stretching occurs slightly higher, around \( \mathrm{3020–3100\ cm^{-1}} \).
- Aldehydes: show characteristic C–H stretching peaks around \( \mathrm{2720–2820\ cm^{-1}} \), often appearing as a weak double peak.
These differences arise due to variations in bond strength and the hybridisation of the carbon atom involved.
(ii) C=C Stretching Absorption (Alkenes)
The carbon–carbon double bond shows a characteristic absorption due to stretching of the \( \mathrm{C=C} \) bond.
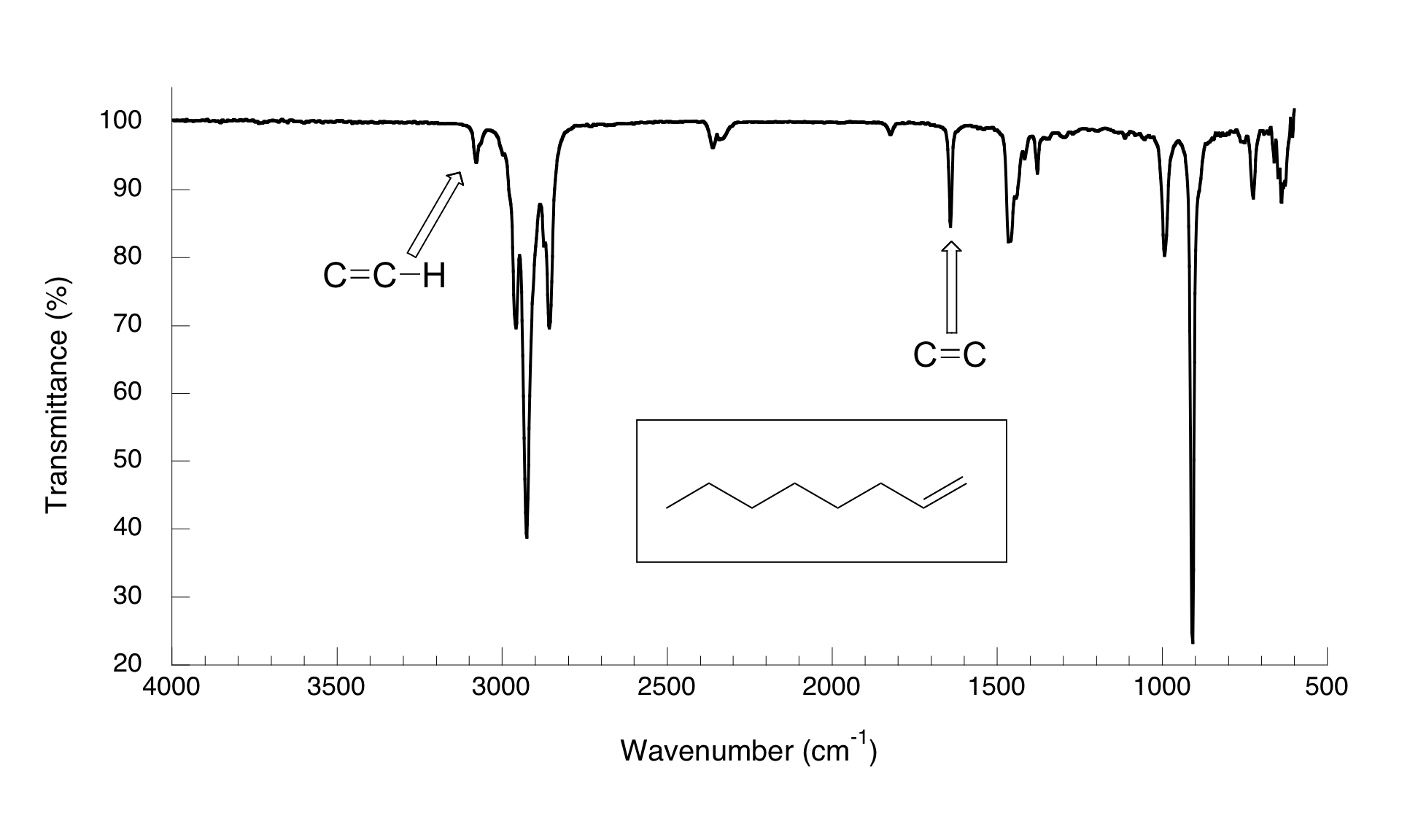
- Occurs at approximately \( \mathrm{1620–1680\ cm^{-1}} \).
- The peak is usually of medium intensity.
- Confirms the presence of a double bond in the molecule.
The exact position may vary slightly depending on substitution and conjugation effects.
(iii) O–H Stretching Absorptions
The O–H bond produces a broad absorption due to hydrogen bonding.
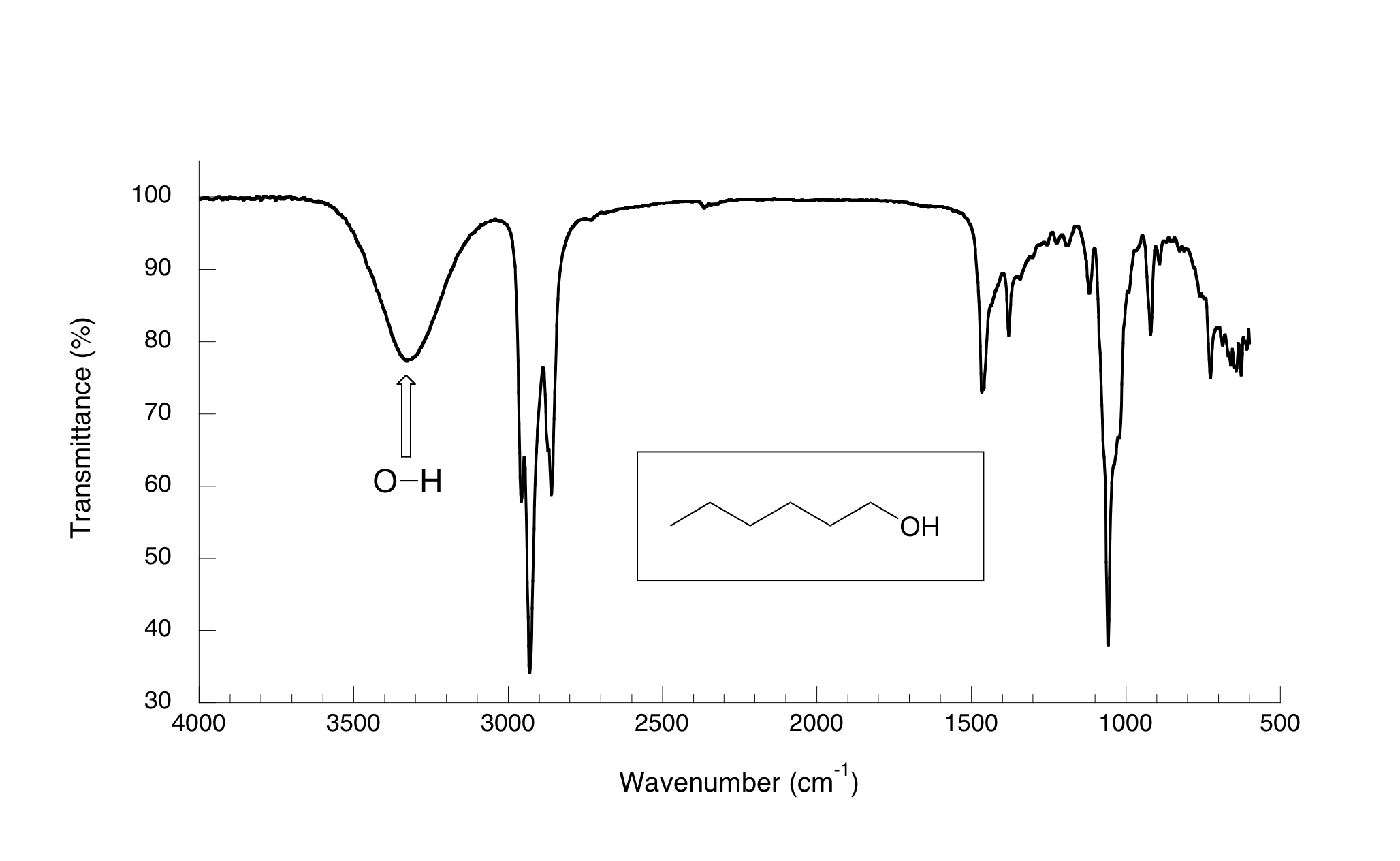
- Alcohols: broad absorption at approximately \( \mathrm{3200–3600\ cm^{-1}} \).
- Carboxylic acids: very broad absorption at approximately \( \mathrm{2500–3300\ cm^{-1}} \), often overlapping with C–H absorptions.
The broadness of the peak is due to intermolecular hydrogen bonding, which causes a range of bond energies and therefore a spread of absorption frequencies.
Key Features for Interpretation
- C–H stretching helps distinguish between alkanes, alkenes and aldehydes.
- The presence of a peak around \( \mathrm{1620–1680\ cm^{-1}} \) indicates a \( \mathrm{C=C} \) bond.
- Broad peaks in the \( \mathrm{2500–3600\ cm^{-1}} \) region indicate O–H groups.
- Peak shape (broad vs sharp) is important for identifying hydrogen bonding.
Example 1:
An IR spectrum shows a broad absorption at \( \mathrm{3300\ cm^{-1}} \) and no peak around \( \mathrm{1700\ cm^{-1}} \). Suggest the functional group present and explain your reasoning.
▶️ Answer/Explanation
The broad absorption around \( \mathrm{3300\ cm^{-1}} \) indicates the presence of an O–H group.
The absence of a peak around \( \mathrm{1700\ cm^{-1}} \) shows that there is no carbonyl group present.
Therefore, the compound is likely an alcohol rather than a carboxylic acid.
Example 2:
A compound shows peaks at \( \mathrm{1650\ cm^{-1}} \) and \( \mathrm{3050\ cm^{-1}} \). Deduce the functional group present and explain how both peaks support your answer.
▶️ Answer/Explanation
The peak at \( \mathrm{1650\ cm^{-1}} \) indicates the presence of a \( \mathrm{C=C} \) double bond.
The peak at \( \mathrm{3050\ cm^{-1}} \) corresponds to C–H stretching in an alkene.
Together, these peaks confirm that the compound contains an alkene functional group.
Infrared spectroscopy allows identification of functional groups based on characteristic absorptions. Certain bonds produce strong, distinctive peaks that are particularly useful in distinguishing between different classes of organic compounds.
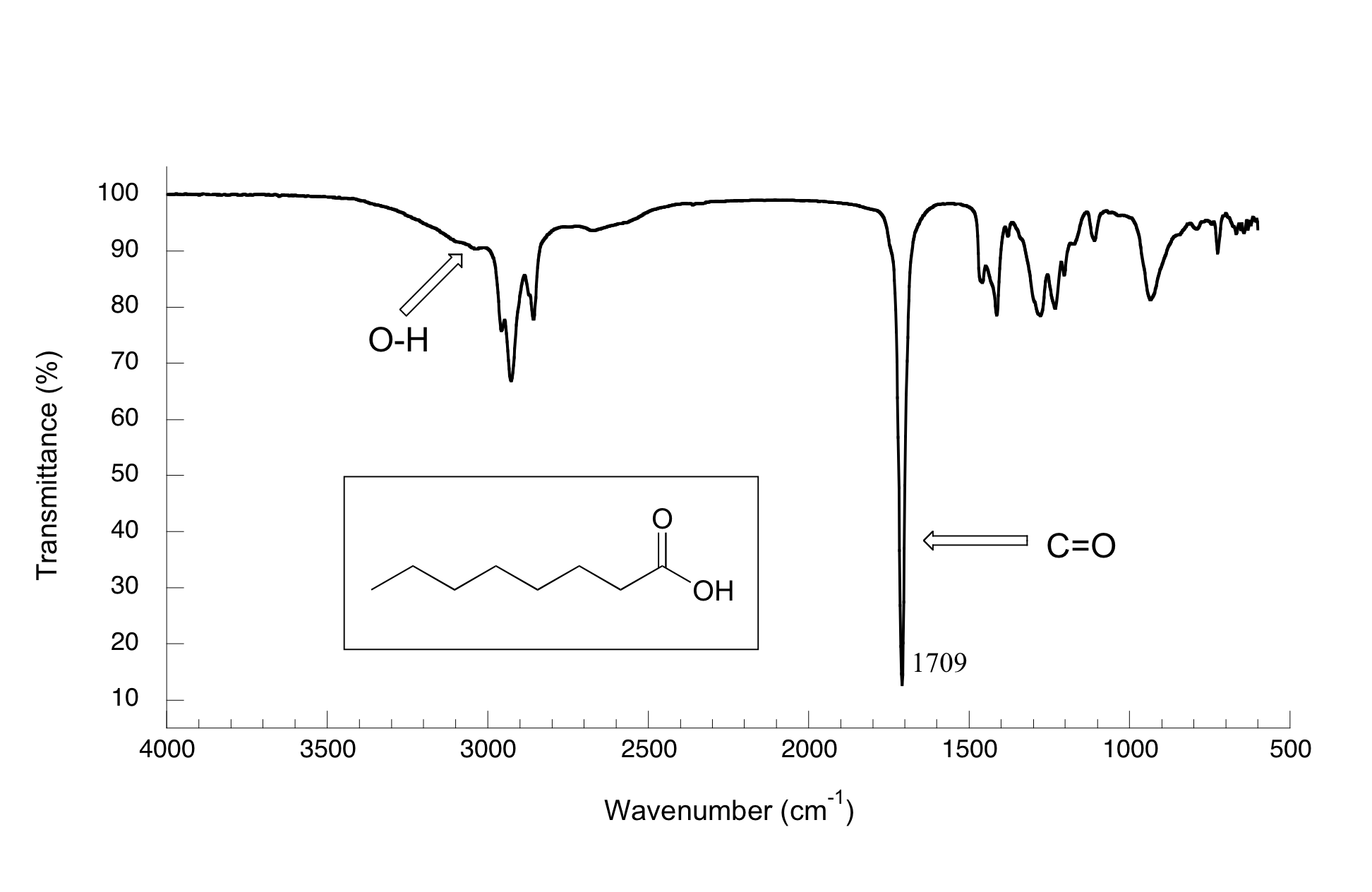
(iv) C=O Stretching Absorptions
The carbon–oxygen double bond produces a strong and sharp absorption due to stretching of the \( \mathrm{C=O} \) bond.
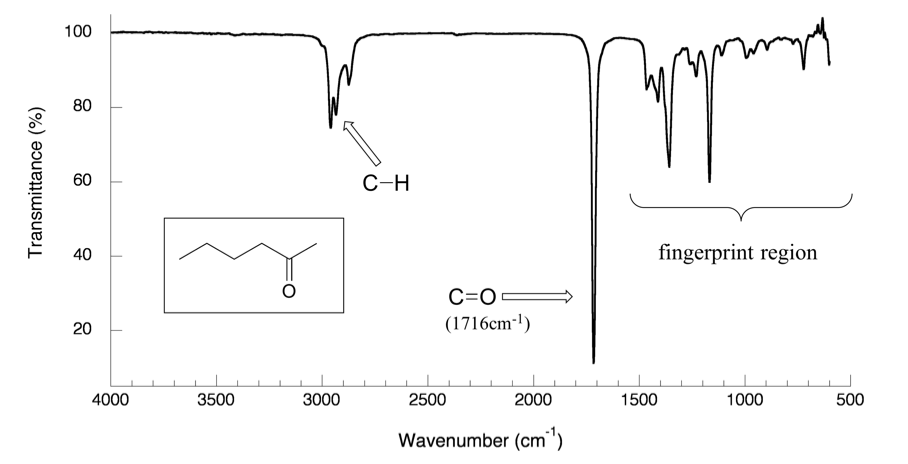
- Occurs at approximately \( \mathrm{1680–1750\ cm^{-1}} \).
- The peak is typically strong and sharp, making it easy to identify.
- Aldehydes: show a \( \mathrm{C=O} \) peak along with characteristic C–H peaks around \( \mathrm{2720–2820\ cm^{-1}} \).
- Ketones: show a \( \mathrm{C=O} \) peak but no aldehyde C–H peaks.
- Carboxylic acids: show a \( \mathrm{C=O} \) peak together with a very broad O–H absorption.
The presence of a strong peak in this region is a key indicator of a carbonyl-containing functional group.
(v) C–X Stretching Absorption (Halogenoalkanes)
Carbon–halogen bonds absorb in the lower wavenumber region due to their lower bond strength.
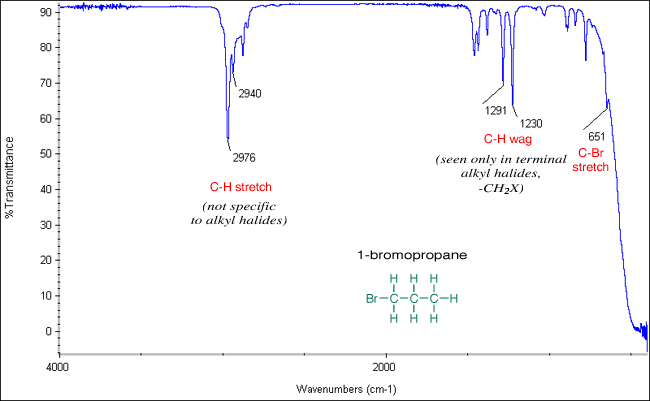
- Occurs at approximately \( \mathrm{500–800\ cm^{-1}} \).
- The exact position depends on the halogen (C–Cl, C–Br, C–I).
- Peaks in this region help identify halogenoalkanes.
These absorptions are often found in the fingerprint region and must be interpreted carefully alongside other peaks.
(vi) N–H Stretching Absorption (Amines)
The nitrogen–hydrogen bond produces a characteristic absorption due to stretching vibrations.
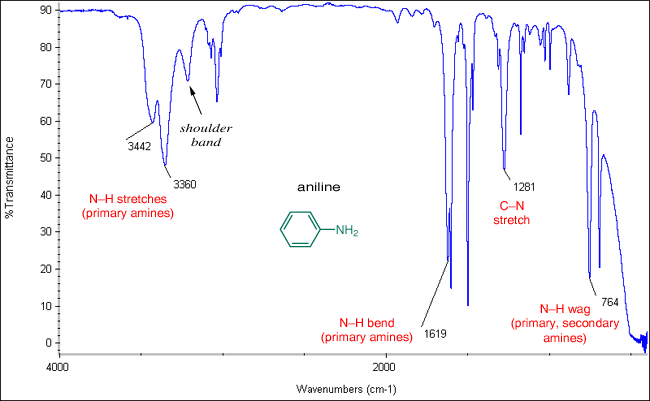
- Occurs at approximately \( \mathrm{3300–3500\ cm^{-1}} \).
- Peaks are usually sharp compared to O–H absorptions.
- Primary amines may show two peaks, while secondary amines show one.
The presence of N–H absorption indicates an amine functional group and can be distinguished from O–H by its sharper peak.
Key Features for Interpretation
- A strong peak around \( \mathrm{1700\ cm^{-1}} \) indicates a carbonyl group.
- Peaks in the \( \mathrm{500–800\ cm^{-1}} \) region suggest a halogen substituent.
- Sharp peaks around \( \mathrm{3300–3500\ cm^{-1}} \) indicate N–H bonds.
- Functional groups are identified by combining multiple peaks rather than relying on a single absorption.
Example 1:
An IR spectrum shows a strong peak at \( \mathrm{1720\ cm^{-1}} \) and no broad O–H absorption. Suggest the functional group present and explain your reasoning.
▶️ Answer/Explanation
The strong peak at \( \mathrm{1720\ cm^{-1}} \) indicates the presence of a carbonyl (\( \mathrm{C=O} \)) group.
The absence of a broad O–H peak shows that the compound is not a carboxylic acid.
Therefore, the compound is likely an aldehyde or a ketone.
Additional peaks (such as aldehyde C–H) would be needed to distinguish between them.
Example 2:
A compound shows a sharp peak at \( \mathrm{3400\ cm^{-1}} \) and absorptions in the region \( \mathrm{600–800\ cm^{-1}} \). Deduce the functional groups present and explain your reasoning.
▶️ Answer/Explanation
The sharp peak at \( \mathrm{3400\ cm^{-1}} \) indicates the presence of an N–H bond, suggesting an amine.
The absorptions in the \( \mathrm{600–800\ cm^{-1}} \) region indicate the presence of a carbon–halogen bond.
Therefore, the compound likely contains both an amine functional group and a halogen substituent.
This conclusion is based on combining evidence from multiple characteristic absorptions.
