Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.23 CORE PRACTICAL 8: Unknown analysis-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.23 CORE PRACTICAL 8: Unknown analysis- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.23 CORE PRACTICAL 8: Unknown analysis- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.23 CORE PRACTICAL 8
Analysis of some inorganic and organic unknowns
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.23 Core Practical 8: Analysis of Inorganic and Organic Unknowns
This practical involves identifying unknown inorganic ions and organic functional groups using qualitative tests. A systematic approach is required, combining observations with known chemical behaviour.
Inorganic Analysis
Test for acids and bases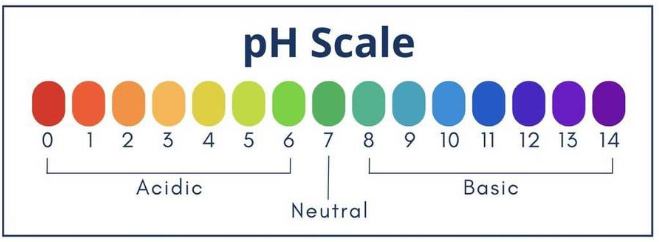

- Blue litmus → red = acid
- Red litmus → blue = base
- Universal indicator used to estimate pH
Test for carbonate ions \( \mathrm{CO_3^{2-}} \)
- Add dilute acid → effervescence
- Gas = \( \mathrm{CO_2} \)
- Turns limewater milky (formation of \( \mathrm{CaCO_3} \))
Test for sulfate ions \( \mathrm{SO_4^{2-}} \)
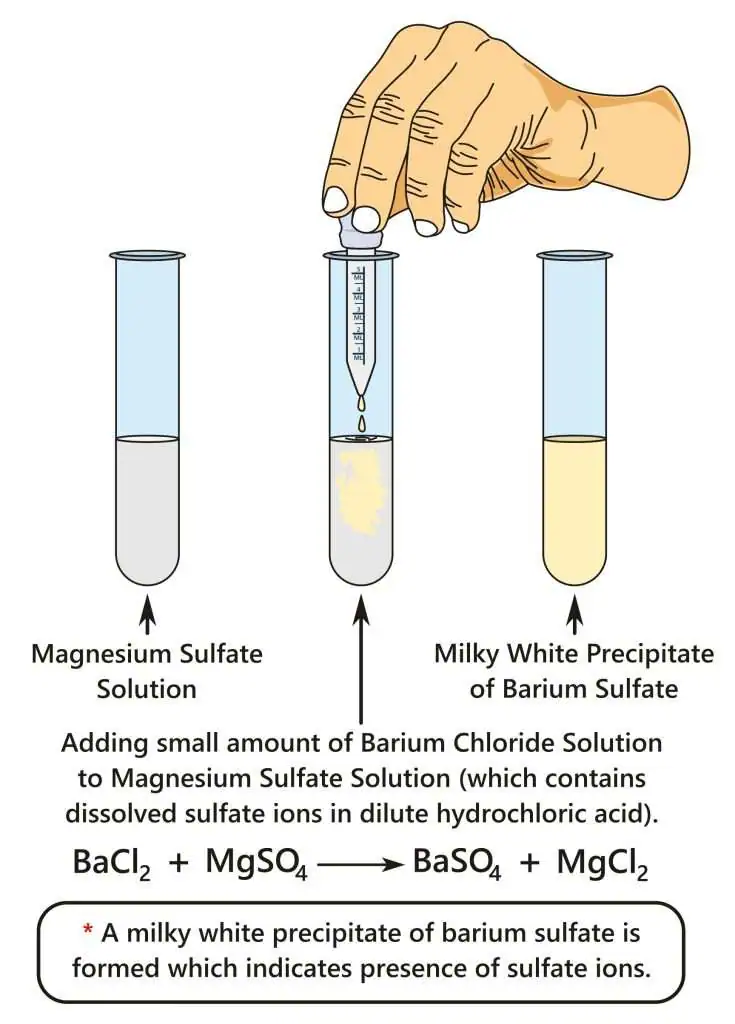
- Add acidified \( \mathrm{Ba^{2+}} \) solution
- White precipitate = \( \mathrm{BaSO_4} \)
- Insoluble in acid
Test for halide ions
- Add dilute \( \mathrm{HNO_3} \) then \( \mathrm{AgNO_3} \)
- \( \mathrm{Cl^-} \): white ppt, soluble in dilute \( \mathrm{NH_3} \)
- \( \mathrm{Br^-} \): cream ppt, soluble in concentrated \( \mathrm{NH_3} \)
- \( \mathrm{I^-} \): yellow ppt, insoluble in \( \mathrm{NH_3} \)
Test for ammonium ions \( \mathrm{NH_4^+} \)
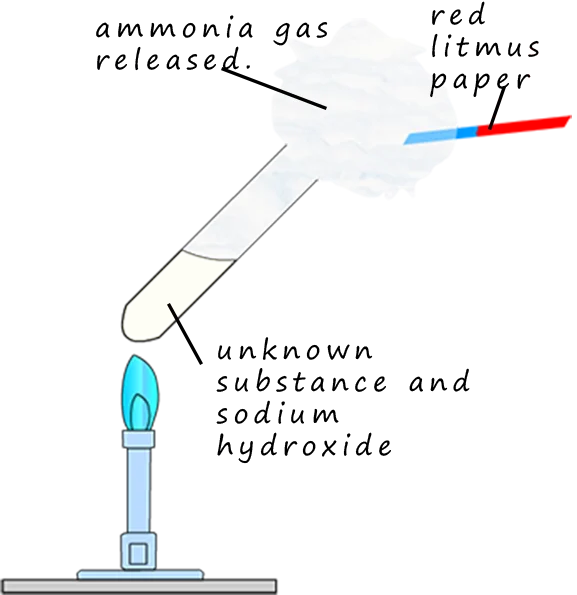
- Add aqueous \( \mathrm{NaOH} \), warm gently
- Ammonia gas released
- Turns damp red litmus paper blue
Test for metal cations (using \( \mathrm{NaOH} \))
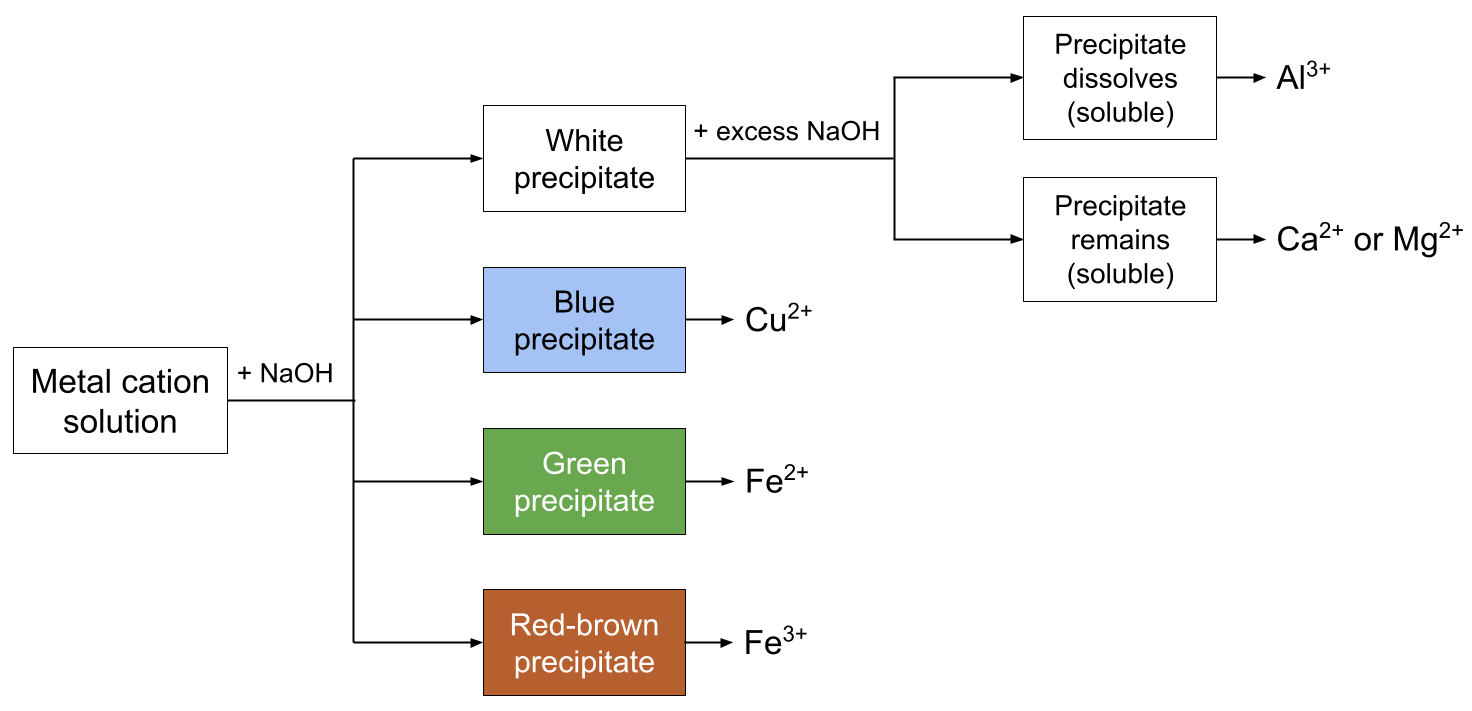
- \( \mathrm{Cu^{2+}} \): blue ppt
- \( \mathrm{Fe^{2+}} \): green ppt (turns brown)
- \( \mathrm{Fe^{3+}} \): brown ppt
- \( \mathrm{Al^{3+}} \), \( \mathrm{Zn^{2+}} \): white ppt (dissolve in excess NaOH)
Organic Analysis
Test for alkenes
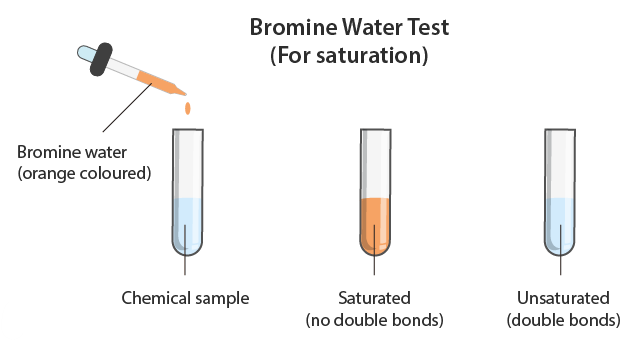
- Add bromine water
- Orange → colourless
Test for alcohols
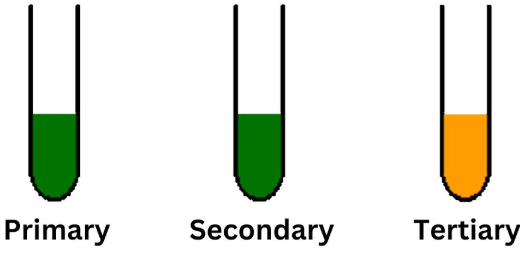
- Add acidified \( \mathrm{K_2Cr_2O_7} \), heat
- Orange → green = primary/secondary alcohol
- No change = tertiary alcohol
Test for carboxylic acids
- Add \( \mathrm{Na_2CO_3} \) or \( \mathrm{NaHCO_3} \)
- Effervescence (CO₂ produced)
Test for aldehydes
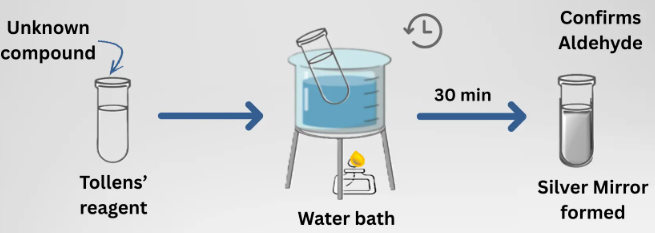
- Tollens’ reagent → silver mirror
- Fehling’s solution → brick-red ppt
Test for ketones
- No reaction with Tollens’ or Fehling’s
Iodoform test
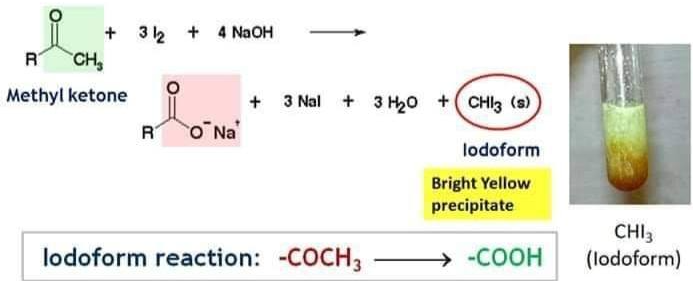
- Warm with iodine + \( \mathrm{NaOH} \)
- Yellow precipitate = \( \mathrm{CHI_3} \)
- Indicates \( \mathrm{CH_3CO-} \) or \( \mathrm{CH_3CH(OH)-} \) group
Solubility behaviour
- Alcohols, amines, acids → may dissolve in water (H-bonding)
- Carboxylic acids dissolve in alkali (form \( \mathrm{RCOO^-} \))
- Amines dissolve in acid (form \( \mathrm{RNH_3^+} \))
Example 1:
An unknown salt produces effervescence with acid and forms a white precipitate with silver nitrate soluble in dilute ammonia.
▶️ Answer
Effervescence → \( \mathrm{CO_3^{2-}} \)
White ppt soluble in NH₃ → \( \mathrm{Cl^-} \)
Salt contains carbonate and chloride ions.
Example 2:
An organic compound decolourises bromine water and gives a positive iodoform test.
▶️ Answer
Decolourisation → alkene present
Iodoform positive → \( \mathrm{CH_3CO-} \) or \( \mathrm{CH_3CH(OH)-} \)
Compound likely contains both alkene and methyl ketone/secondary alcohol structure.
