Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.5 Bond polarity and mechanism type-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.5 Bond polarity and mechanism type- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.5 Bond polarity and mechanism type- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.5 understand the link between bond polarity and the type of reaction mechanism a compound will undergo
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.5 Bond Polarity and Type of Reaction Mechanism
The type of reaction mechanism that an organic compound undergoes is strongly influenced by the polarity of its bonds. Bond polarity arises due to differences in electronegativity between atoms, leading to uneven distribution of electron density. This creates partial charges within molecules, which determine how they interact with electrophiles and nucleophiles.
Bond Polarity
A polar bond is a covalent bond in which electrons are shared unequally, resulting in a partial positive charge (\( \delta^+ \)) and a partial negative charge (\( \delta^- \)).

This polarity creates regions of electron deficiency and electron richness within a molecule, making certain atoms more susceptible to attack by nucleophiles or electrophiles.
Effect on Reaction Mechanisms
Polar bonds can undergo heterolytic fission, producing ions that act as electrophiles and nucleophiles.
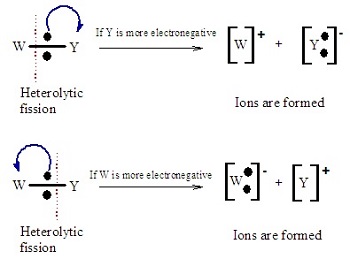
- The direction of polarity determines which atom is electron-deficient and which is electron-rich.
- Electron-deficient centres (\( \delta^+ \)) are attacked by nucleophiles.
- Electron-rich regions (\( \delta^- \)) can attract electrophiles.
Example: Haloalkanes
\( \mathrm{C-Br} \) bond is polar because bromine is more electronegative than carbon.
- Carbon has a partial positive charge (\( \delta^+ \)).
- Bromine has a partial negative charge (\( \delta^- \)).
- The bond can break heterolytically to form \( \mathrm{C^+} \) and \( \mathrm{Br^-} \).
- The \( \delta^+ \) carbon is attacked by nucleophiles.
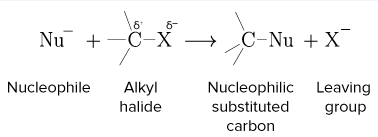
Therefore, haloalkanes typically undergo nucleophilic substitution reactions.
Example: Alkenes
\( \mathrm{C=C} \) double bond is a region of high electron density due to the π bond.
- The π bond is electron-rich and easily attacked.
- Electrophiles are attracted to this region.
- The π bond breaks to form new single bonds.
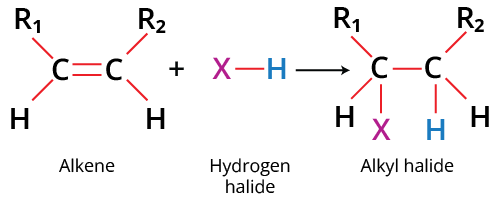
Therefore, alkenes typically undergo electrophilic addition reactions.
Link Between Polarity and Mechanism
- Polar bonds favour reactions involving ions (heterolytic mechanisms).
- Electron-deficient centres determine sites of nucleophilic attack.
- Electron-rich regions determine sites of electrophilic attack.
- The type of bond present helps predict the reaction mechanism.
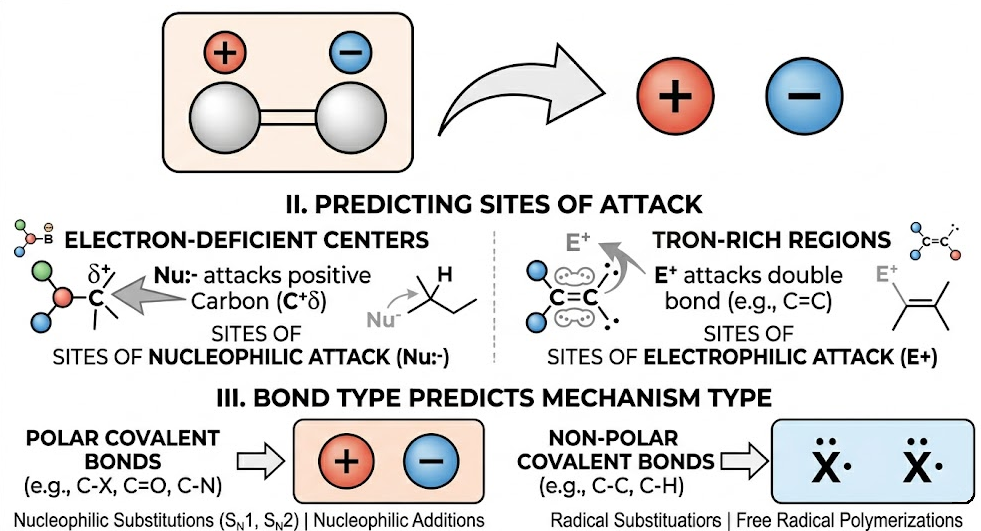
Thus, understanding bond polarity allows prediction of whether a compound is more likely to undergo substitution, addition, or other types of reactions.
Example 1:
Explain why haloalkanes undergo nucleophilic substitution reactions.
▶️ Answer/Explanation
The \( \mathrm{C-X} \) bond in haloalkanes is polar because the halogen is more electronegative than carbon.
This creates a partial positive charge on the carbon atom, making it electron-deficient.
Nucleophiles are attracted to this \( \delta^+ \) carbon and donate a pair of electrons to form a new bond.
The bond breaks heterolytically, leading to substitution of the halogen.
Example 2:
Explain why alkenes undergo electrophilic addition rather than nucleophilic substitution.
▶️ Answer/Explanation
Alkenes contain a \( \mathrm{C=C} \) double bond with a π bond that is rich in electron density.
This electron-rich region attracts electrophiles rather than nucleophiles.
The electrophile accepts an electron pair from the π bond, breaking the double bond and forming new single bonds.
Therefore, alkenes undergo electrophilic addition instead of substitution.
