Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.6 Nomenclature and structures-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.6 Nomenclature and structures- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.6 Nomenclature and structures- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.6 understand the nomenclature of halogenoalkanes and be able to draw their structural, displayed and skeletal formulae
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.6 Nomenclature and Representation of Halogenoalkanes
Halogenoalkanes are organic compounds in which one or more hydrogen atoms in an alkane have been replaced by halogen atoms such as fluorine, chlorine, bromine or iodine. Understanding their nomenclature and different ways of representing their structures is essential for identifying compounds and predicting their chemical behaviour.
Nomenclature of Halogenoalkanes
Halogenoalkanes are named by identifying the longest carbon chain and naming the halogen substituents as prefixes.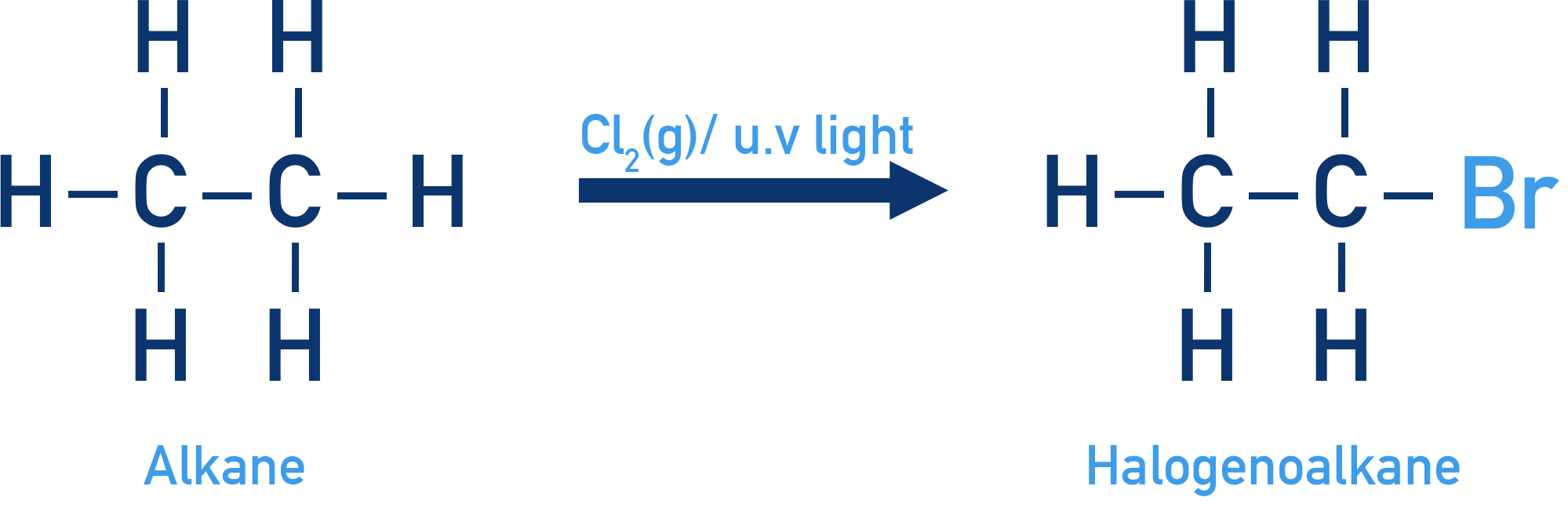
- The parent chain is named as an alkane (methane, ethane, propane, etc.).
- Halogen atoms are named as prefixes: fluoro-, chloro-, bromo-, iodo-.
- The position of the halogen is indicated by a number.
- Numbering is done to give the substituents the lowest possible numbers.
- If more than one halogen is present, prefixes such as di-, tri- are used.
- Substituents are listed in alphabetical order.
Example: \( \mathrm{CH_3CHClCH_3} \) → 2-chloropropane
Structural Formula
A structural formula shows the arrangement of atoms in a molecule but does not display all bonds explicitly.
- Groups of atoms are written together.
- Does not show all individual bonds.
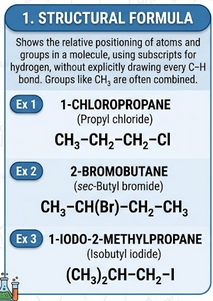
Example: \( \mathrm{CH_3CH_2Cl} \)
Displayed Formula
A displayed formula shows all atoms and all bonds in a molecule.
- Every bond is drawn explicitly.
- Useful for showing bonding and structure clearly.
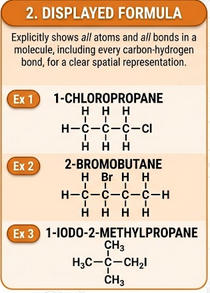
Example (chloroethane):
Skeletal Formula
A skeletal formula represents the carbon framework as lines, omitting hydrogen atoms attached to carbon.
- Each line represents a carbon-carbon bond.
- Carbon atoms are implied at the ends and intersections of lines.
- Hydrogen atoms attached to carbon are not shown.
- Functional groups (e.g. Cl, Br) are shown explicitly.
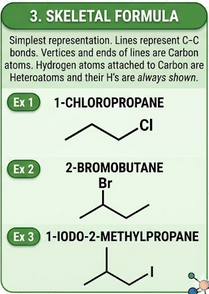
Example: chloroethane is represented as a two-carbon line with Cl attached.
Key Points
- Correct naming depends on identifying the longest carbon chain and substituent positions.
- Different representations show different levels of structural detail.
- Structural formula is compact, displayed formula is detailed, and skeletal formula is simplified.
Example 1:
Name the compound \( \mathrm{CH_3CH_2CH_2Br} \) and explain your reasoning.
▶️ Answer/Explanation
The longest carbon chain contains three carbon atoms, so the parent name is propane.
A bromine atom is attached to one end of the chain, giving it position 1.
Therefore, the compound is named 1-bromopropane.
Example 2:
Draw and explain the skeletal and displayed formulae of 2-chloropropane.
▶️ Answer/Explanation
The compound has a three-carbon chain (propane) with a chlorine atom on the second carbon.
In the displayed formula, all atoms and bonds are shown, including the \( \mathrm{C-Cl} \) bond on the middle carbon.
In the skeletal formula, the three-carbon chain is represented as a V-shaped line, and the chlorine atom is shown attached to the middle vertex.
Hydrogen atoms bonded to carbon are not shown in the skeletal formula but are implied.
