Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.7 Primary, secondary and tertiary halogenoalkanes-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.7 Primary, secondary and tertiary halogenoalkanes- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.7 Primary, secondary and tertiary halogenoalkanes- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.7 understand the distinction between primary, secondary and tertiary halogenoalkanes
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.7 Primary, Secondary and Tertiary Halogenoalkanes
Halogenoalkanes can be classified as primary, secondary or tertiary depending on the number of alkyl groups attached to the carbon atom bonded to the halogen. This classification is important because it influences the reactivity and the type of reaction mechanism the compound undergoes.
Classification of Halogenoalkanes
The classification depends on how many carbon atoms are directly attached to the carbon bearing the halogen.
- Primary (1°) halogenoalkane: the carbon bonded to the halogen is attached to only one other carbon atom.
- Secondary (2°) halogenoalkane: the carbon bonded to the halogen is attached to two other carbon atoms.
- Tertiary (3°) halogenoalkane: the carbon bonded to the halogen is attached to three other carbon atoms.
Structural Representation
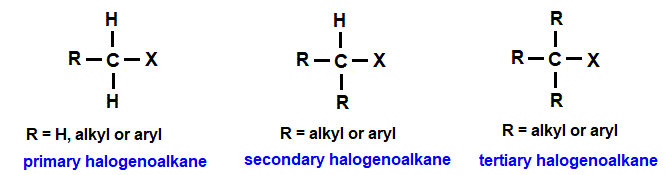
- Primary: \( \mathrm{R-CH_2-X} \)
- Secondary: \( \mathrm{R-CH(X)-R’} \)
- Tertiary: \( \mathrm{R-C(X)(R’)(R”)} \)
Explanation of the Distinction
- The classification reflects the degree of substitution of the carbon atom bonded to the halogen.
- More substituted carbons (tertiary) have more alkyl groups attached, increasing electron density around the carbon.
- This affects both the stability of intermediates (such as carbocations) and the steric hindrance around the reactive centre.
Effect on Reactivity
- Tertiary halogenoalkanes form more stable carbocations due to the electron-donating effect of alkyl groups.
- Primary halogenoalkanes have less steric hindrance, making nucleophilic attack easier.
- Secondary halogenoalkanes show intermediate behaviour.
Therefore, the classification helps predict whether a compound is more likely to undergo mechanisms such as \( \mathrm{S_N1} \) or \( \mathrm{S_N2} \).
Example 1:
Classify the compound \( \mathrm{CH_3CH_2CH_2Cl} \) and justify your answer.
▶️ Answer/Explanation
The carbon bonded to chlorine is attached to only one other carbon atom.
Therefore, it is a primary halogenoalkane.
This means it has low steric hindrance and is more likely to undergo nucleophilic substitution via an \( \mathrm{S_N2} \) mechanism.
Example 2:
Explain why tertiary halogenoalkanes are more likely to undergo \( \mathrm{S_N1} \) reactions than primary halogenoalkanes.
▶️ Answer/Explanation
Tertiary halogenoalkanes form tertiary carbocations when the \( \mathrm{C-X} \) bond breaks heterolytically.
These carbocations are stabilised by the electron-donating effect of three alkyl groups.
In contrast, primary carbocations are much less stable and do not form easily.
Therefore, tertiary halogenoalkanes favour the \( \mathrm{S_N1} \) mechanism, which involves carbocation formation.
