Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.8 Reactions of halogenoalkanes-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.8 Reactions of halogenoalkanes- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.8 Reactions of halogenoalkanes- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.8 understand the reactions of halogenoalkanes with:
i aqueous alkali, including KOH(aq) to produce alcohols (where the hydroxide ion acts as a nucleophile)
ii ethanolic potassium hydroxide to produce alkenes by an elimination reaction (where the hydroxide ion acts as a base)
iii aqueous silver nitrate in ethanol (where water acts as a nucleophile)
iv alcoholic ammonia under pressure to produce amines (where the ammonia acts as a nucleophile)
v alcoholic potassium cyanide to produce nitriles (where the cyanide ion acts as a nucleophile)
Students should know this is an example of increasing the length of the carbon chain
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.8 Reactions of Halogenoalkanes (i–iii)
Halogenoalkanes undergo a variety of reactions depending on the conditions and reagents used. These reactions involve either nucleophilic substitution or elimination, and the role of the reagent (as a nucleophile or base) determines the mechanism and products formed.
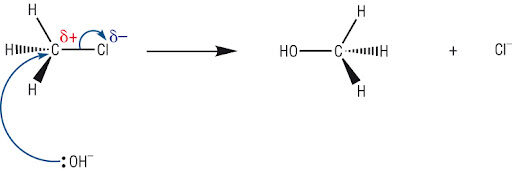
(i) Reaction with Aqueous Alkali (KOH(aq))
Halogenoalkanes react with aqueous potassium hydroxide to form alcohols via nucleophilic substitution.
- The \( \mathrm{OH^-} \) ion acts as a nucleophile.
- It donates a lone pair of electrons to the \( \delta^+ \) carbon bonded to the halogen.
- The \( \mathrm{C-X} \) bond breaks heterolytically, forming a halide ion.
- The halogen is replaced by a hydroxyl group (\( \mathrm{-OH} \)).
(ii) Reaction with Ethanolic Potassium Hydroxide (KOH in ethanol)
Halogenoalkanes react with ethanolic potassium hydroxide to form alkenes via elimination.
- The \( \mathrm{OH^-} \) ion acts as a base.
- It removes a hydrogen atom from a carbon adjacent to the \( \mathrm{C-X} \) bond.
- The electrons from the \( \mathrm{C-H} \) bond form a \( \mathrm{C=C} \) double bond.
- The halogen leaves as a halide ion.
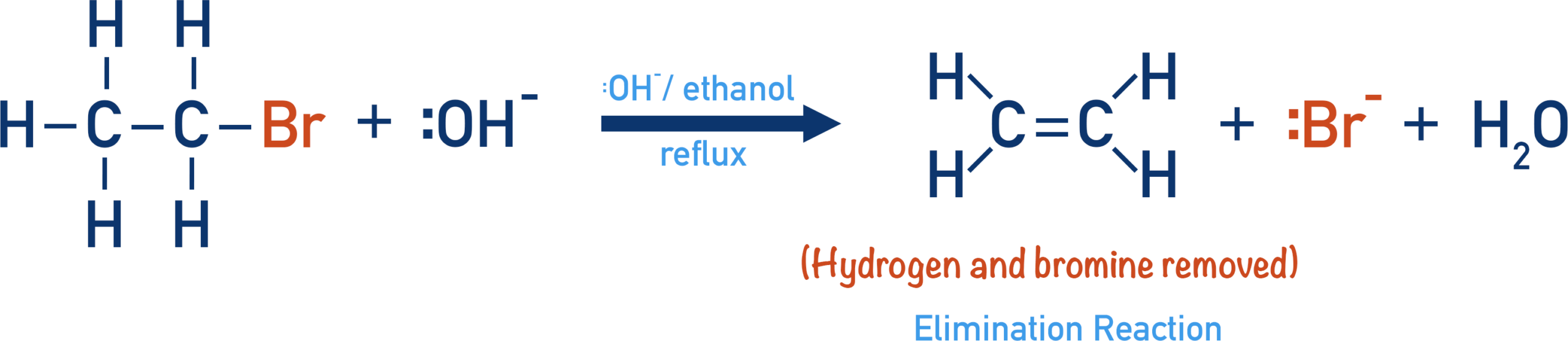
This is an elimination reaction, producing an alkene and a hydrogen halide.
(iii) Reaction with Aqueous Silver Nitrate in Ethanol
Halogenoalkanes react with aqueous silver nitrate in ethanol to undergo hydrolysis, forming alcohols.
- Water acts as a nucleophile.
- The \( \mathrm{C-X} \) bond breaks heterolytically to form a carbocation.
- Water donates a lone pair to the carbocation to form an alcohol.
- The halide ion reacts with \( \mathrm{Ag^+} \) to form a precipitate.
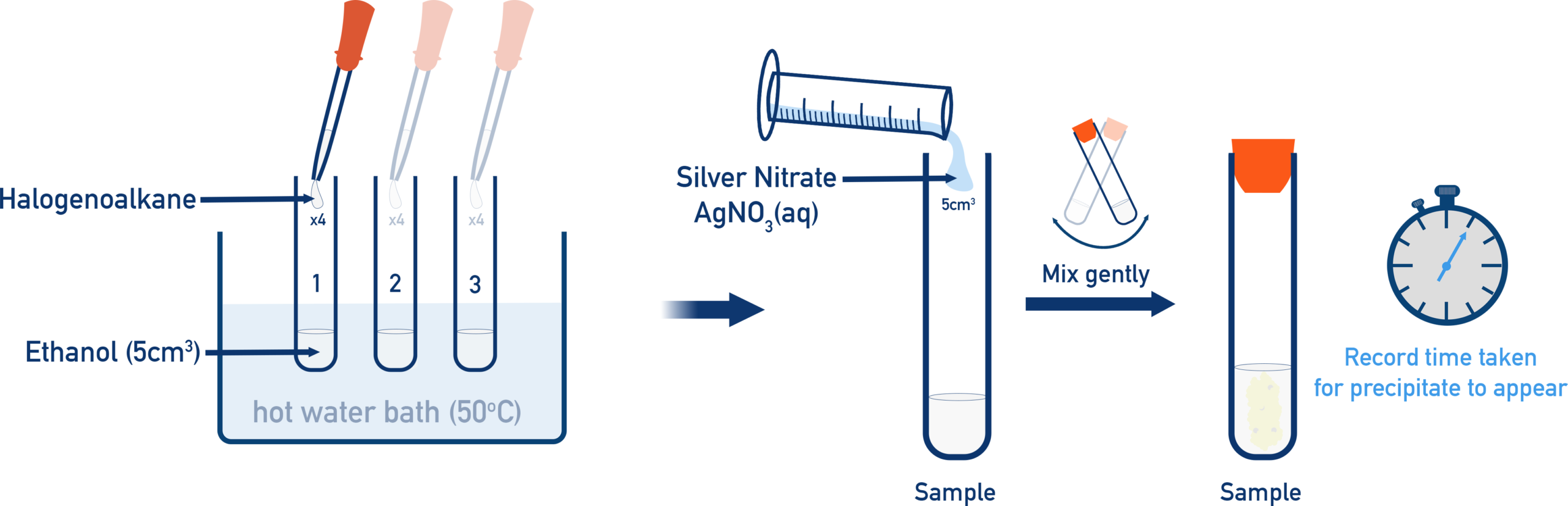
Example: \( \mathrm{CH_3CH_2Cl + H_2O \rightarrow CH_3CH_2OH + HCl} \)
- \( \mathrm{AgCl} \) forms a white precipitate.
- This reaction is used to test for halogenoalkanes and compare their reactivity.
Key Comparisons
- Aqueous KOH favours substitution (formation of alcohols).
- Ethanolic KOH favours elimination (formation of alkenes).
- Silver nitrate reaction involves hydrolysis and formation of a precipitate.
- The role of \( \mathrm{OH^-} \) changes: nucleophile in (i), base in (ii).
Example 1:
Explain why aqueous KOH produces an alcohol whereas ethanolic KOH produces an alkene when reacting with a halogenoalkane.
▶️ Answer/Explanation
In aqueous KOH, the \( \mathrm{OH^-} \) ion is surrounded by water molecules and acts primarily as a nucleophile.
It attacks the electron-deficient carbon and substitutes the halogen, forming an alcohol.
In ethanolic KOH, the \( \mathrm{OH^-} \) ion acts as a base and removes a hydrogen atom from a neighbouring carbon.
This leads to elimination and formation of a \( \mathrm{C=C} \) double bond, producing an alkene.
Example 2:
Explain how aqueous silver nitrate in ethanol can be used to compare the reactivity of halogenoalkanes.
▶️ Answer/Explanation
When halogenoalkanes react with aqueous silver nitrate, the \( \mathrm{C-X} \) bond breaks and halide ions are released.
These halide ions react with \( \mathrm{Ag^+} \) to form a precipitate such as \( \mathrm{AgCl} \), \( \mathrm{AgBr} \), or \( \mathrm{AgI} \).
The rate at which the precipitate forms indicates the rate of hydrolysis.
More reactive halogenoalkanes (e.g. tertiary) form precipitates faster than less reactive ones (e.g. primary).
10.8 Reactions of Halogenoalkanes (iv–v)
Halogenoalkanes can undergo further nucleophilic substitution reactions with different nucleophiles, leading to the formation of new functional groups. These reactions are important in organic synthesis as they allow the conversion of halogenoalkanes into more complex molecules, including amines and nitriles.
(iv) Reaction with Alcoholic Ammonia (NH₃ in ethanol, under pressure)
Halogenoalkanes react with alcoholic ammonia to form amines via nucleophilic substitution.
- \( \mathrm{NH_3} \) acts as a nucleophile due to its lone pair of electrons.
- It attacks the \( \delta^+ \) carbon bonded to the halogen.
- The \( \mathrm{C-X} \) bond breaks heterolytically, forming a halide ion.
- An intermediate ammonium ion is formed, which then loses a proton.
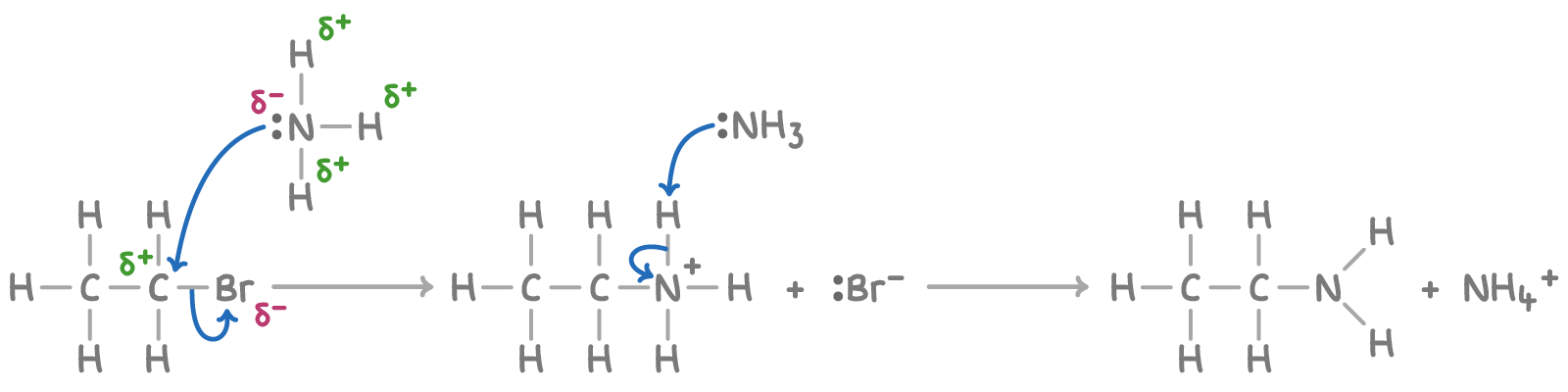
- Excess ammonia is used to favour formation of the primary amine.
- Reaction is carried out under pressure to keep ammonia in solution.
(v) Reaction with Alcoholic Potassium Cyanide (KCN in ethanol)
Halogenoalkanes react with alcoholic potassium cyanide to form nitriles via nucleophilic substitution.
- \( \mathrm{CN^-} \) acts as a nucleophile.
- It donates a lone pair of electrons to the \( \delta^+ \) carbon.
- The \( \mathrm{C-X} \) bond breaks heterolytically.
- A nitrile group (\( \mathrm{-C \equiv N} \)) is formed.
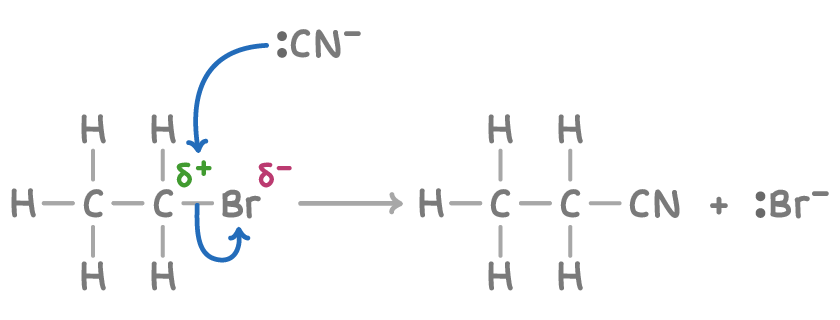
Extension of Carbon Chain
- The nitrile group introduces an additional carbon atom into the molecule.
- This increases the length of the carbon chain by one carbon.
- This reaction is important in synthesis for building larger organic molecules.
Key Comparisons
- Both reactions are nucleophilic substitution.
- Different nucleophiles produce different functional groups (amines vs nitriles).
- Reaction with \( \mathrm{CN^-} \) increases carbon chain length, while \( \mathrm{NH_3} \) does not.
- Conditions (ethanol, pressure, excess reagent) influence the product formed.
Example 1:
Explain why excess ammonia is used in the reaction of halogenoalkanes with ammonia.
▶️ Answer/Explanation
Ammonia reacts with halogenoalkanes to form primary amines, but the amine product can also act as a nucleophile and react further.
This can lead to the formation of secondary and tertiary amines.
Using excess ammonia increases the likelihood that ammonia, rather than the amine product, reacts with the halogenoalkane.
This favours the formation of the primary amine as the main product.
Example 2:
Explain how the reaction of a halogenoalkane with potassium cyanide can be used to increase the carbon chain length.
▶️ Answer/Explanation
The \( \mathrm{CN^-} \) ion acts as a nucleophile and replaces the halogen atom in the halogenoalkane.
The nitrile group formed contains an additional carbon atom compared to the original molecule.
This results in an increase in the carbon chain length by one carbon atom.
Therefore, this reaction is useful for synthesising longer-chain organic compounds.
