Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 10.9 Nucleophilic substitution mechanisms-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.9 Nucleophilic substitution mechanisms- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 10.9 Nucleophilic substitution mechanisms- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
10.9 understand the mechanisms of the nucleophilic substitution reactions between primary halogenoalkanes and:
i aqueous potassium hydroxide
ii ammonia
SN1 and SN2 substitution mechanisms will be tested in Unit 4
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
10.9 Nucleophilic Substitution Mechanisms of Primary Halogenoalkanes
Primary halogenoalkanes undergo nucleophilic substitution reactions via a specific mechanism known as the \( \mathrm{S_N2} \) mechanism. This mechanism involves a single step in which bond breaking and bond formation occur simultaneously. The nature of the nucleophile and reaction conditions determines the product formed.
SN2 Mechanism
The \( \mathrm{S_N2} \) mechanism is a one-step nucleophilic substitution in which the nucleophile attacks the carbon atom as the leaving group departs.
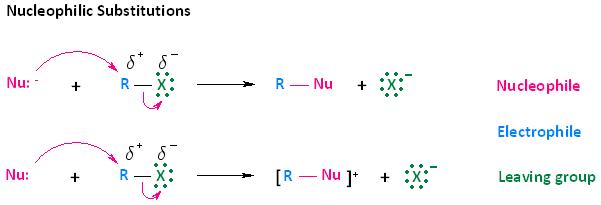
- Occurs mainly in primary halogenoalkanes due to low steric hindrance.
- Involves a single transition state (no intermediate).
- Bond formation and bond breaking occur simultaneously.
- The nucleophile attacks from the opposite side (backside attack).
(i) Reaction with Aqueous Potassium Hydroxide
Primary halogenoalkanes react with aqueous KOH to form alcohols via an \( \mathrm{S_N2} \) mechanism.

- \( \mathrm{OH^-} \) acts as a strong nucleophile.
- It attacks the \( \delta^+ \) carbon from the opposite side of the halogen.
- The \( \mathrm{C-X} \) bond breaks heterolytically as the new \( \mathrm{C-OH} \) bond forms.
- A halide ion is released.
Example: \( \mathrm{CH_3CH_2Br + OH^- \rightarrow CH_3CH_2OH + Br^-} \)
(ii) Reaction with Ammonia
Primary halogenoalkanes react with ammonia to form amines via an \( \mathrm{S_N2} \) mechanism.
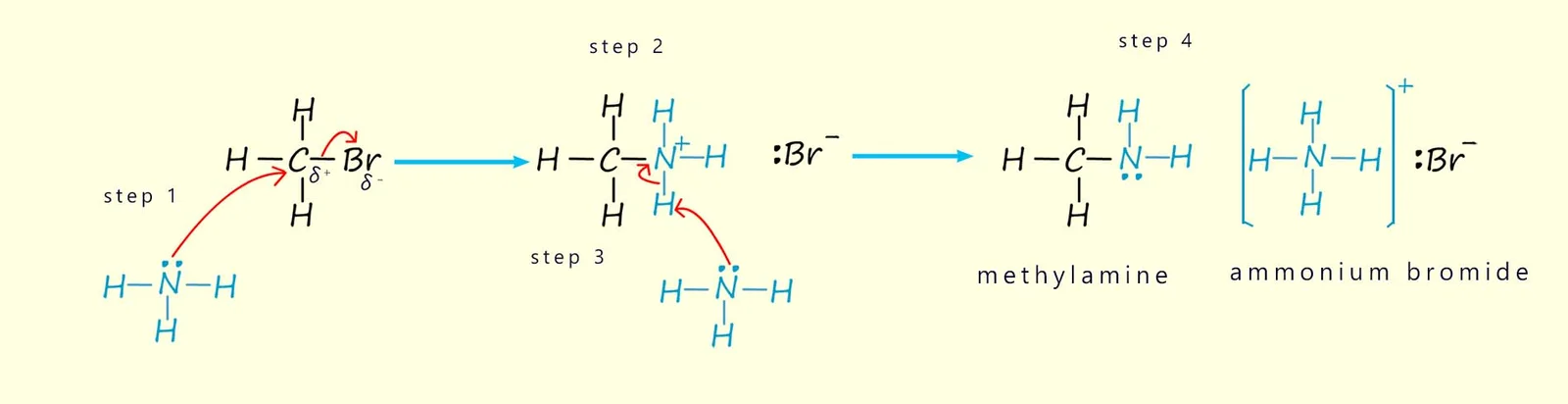
- \( \mathrm{NH_3} \) acts as a nucleophile due to its lone pair.
- It attacks the \( \delta^+ \) carbon in a single step.
- The \( \mathrm{C-X} \) bond breaks as the \( \mathrm{C-N} \) bond forms.
- An intermediate ammonium ion is formed, which then loses a proton to give the amine.
Example: \( \mathrm{CH_3CH_2Br + NH_3 \rightarrow CH_3CH_2NH_2 + HBr} \)
Why Primary Halogenoalkanes Favour SN2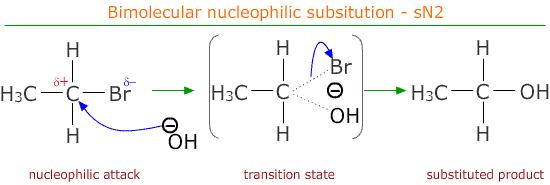
- The carbon atom is less hindered, allowing easy access for the nucleophile.
- There is minimal steric hindrance from surrounding alkyl groups.
- Formation of unstable primary carbocations is avoided, so \( \mathrm{S_N1} \) is not favoured.
Key Features of the Mechanism
- Single-step mechanism with no intermediate.
- Involves simultaneous bond making and breaking.
- Requires a strong nucleophile.
- Rate depends on both the halogenoalkane and nucleophile concentration.
Example 1:
Explain why primary halogenoalkanes undergo nucleophilic substitution via an \( \mathrm{S_N2} \) mechanism rather than an \( \mathrm{S_N1} \) mechanism.
▶️ Answer/Explanation
Primary halogenoalkanes do not readily form carbocations because primary carbocations are highly unstable.
Therefore, the \( \mathrm{S_N1} \) mechanism, which involves carbocation formation, is not favoured.
Instead, the reaction occurs in a single step where the nucleophile attacks the carbon as the leaving group departs.
The low steric hindrance around the carbon allows this simultaneous process, favouring the \( \mathrm{S_N2} \) mechanism.
Example 2:
Describe the mechanism of the reaction between bromoethane and ammonia.
▶️ Answer/Explanation
The ammonia molecule, with a lone pair of electrons, acts as a nucleophile and attacks the \( \delta^+ \) carbon atom in bromoethane from the opposite side of the bromine atom.
As the \( \mathrm{C-N} \) bond forms, the \( \mathrm{C-Br} \) bond breaks heterolytically, releasing a bromide ion.
An intermediate ethylammonium ion is formed, which then loses a proton to produce ethylamine.
This occurs in a single step characteristic of the \( \mathrm{S_N2} \) mechanism.
