Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 6.1 Enthalpy change (ΔH) and standard conditions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.1 Enthalpy change (ΔH) and standard conditions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.1 Enthalpy change (ΔH) and standard conditions- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
6.1 know that the enthalpy change, ΔH, is the heat energy change measured at constant pressure and that standard conditions are 100 kPa and a specified temperature, usually 298 K
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
6.1 Enthalpy Change (ΔH) and Standard Conditions
Enthalpy change (\( \Delta H \)) is a key concept in thermochemistry and refers to the heat energy change during a chemical reaction carried out at constant pressure.
Definition of Enthalpy Change
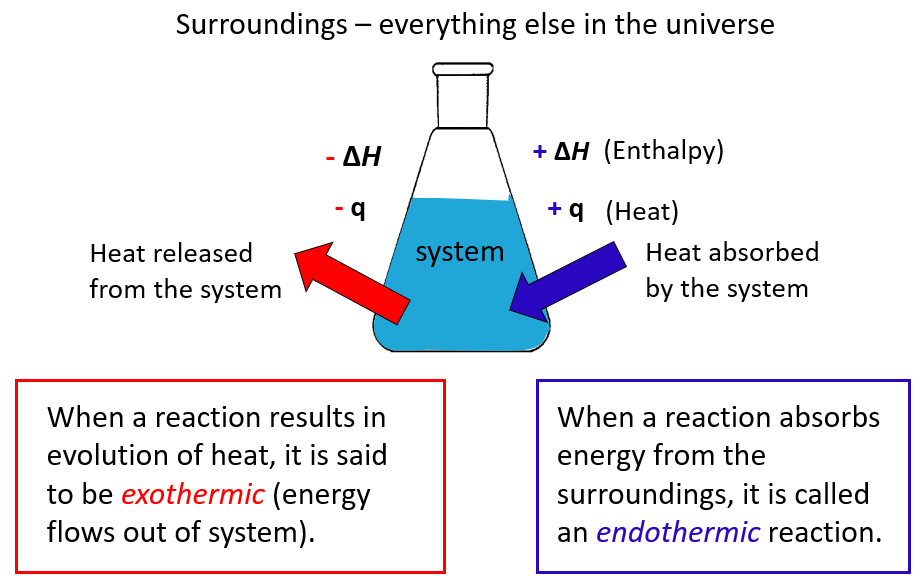
The enthalpy change (\( \Delta H \)) is the heat energy absorbed or released when a reaction occurs at constant pressure.
- Measured in kJ mol⁻¹.
- Depends on the amount of substance reacting.
Sign Convention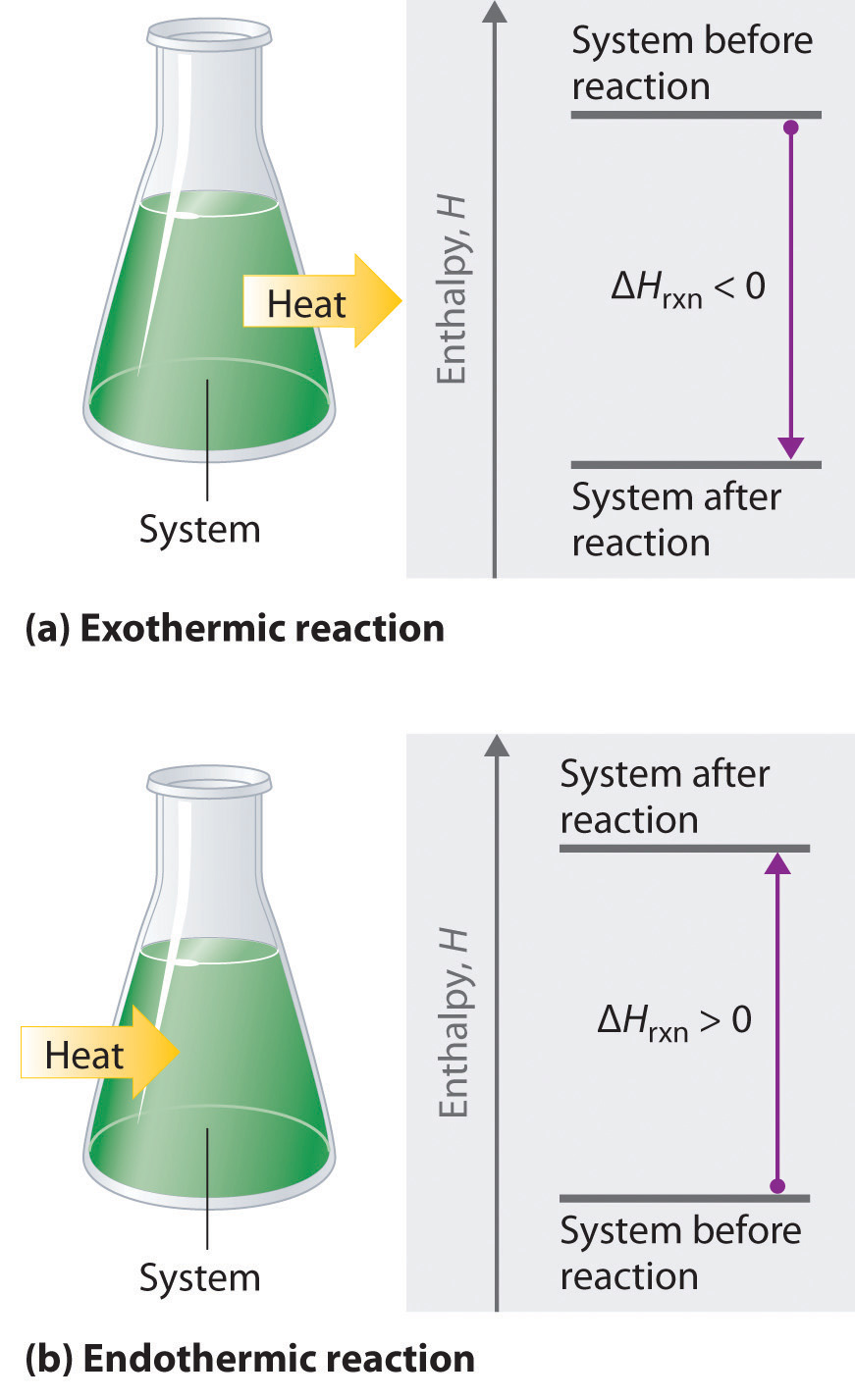
Exothermic reaction:
- Heat is released to surroundings.
- \( \Delta H < 0 \) (negative).
Endothermic reaction:
- Heat is absorbed from surroundings.
- \( \Delta H > 0 \) (positive).
Constant Pressure Condition
- Most reactions are carried out in open containers.
- Pressure remains constant (atmospheric pressure).
- Therefore, heat change measured = enthalpy change.
Standard Conditions

- Pressure = 100 kPa
- Temperature = 298 K (unless otherwise stated)
- Solutions = 1 mol dm⁻³
- Substances in their standard states
Standard State
- Most stable form under standard conditions.
- Examples: \( \mathrm{O_2(g)} \), \( \mathrm{C(graphite)} \), \( \mathrm{H_2(g)} \)
Example of Enthalpy Change
Combustion of methane:
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
This reaction is exothermic, so \( \Delta H < 0 \).
Key Points
- \( \Delta H \) is heat change at constant pressure.
- Standard conditions: 100 kPa, usually 298 K.
- Allows comparison of enthalpy values.
Therefore, enthalpy change provides a standard way to measure energy changes in reactions.
Example 1 :
Explain why \( \Delta H \) is negative for combustion reactions.
▶️ Answer/Explanation
Combustion releases heat to surroundings.
Energy of products is lower than reactants.
Therefore, \( \Delta H \) is negative.
Example 2 :
State standard conditions and explain their importance.
▶️ Answer/Explanation
100 kPa and 298 K.
Ensures consistent comparison of enthalpy values.
