Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 6.11 Bond enthalpy and reaction insights-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.11 Bond enthalpy and reaction insights- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.11 Bond enthalpy and reaction insights- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
6.11 understand that bond enthalpy data gives some indication about which bond will break first in a reaction, how easy or difficult it is and therefore how rapidly a reaction will take place at room temperature
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
6.11 Bond Enthalpy and Reactivity
Bond enthalpy data provides insight into which bonds are most likely to break during a reaction and therefore helps predict reactivity and reaction rate.
Key Idea
- Bond enthalpy = energy required to break a bond.
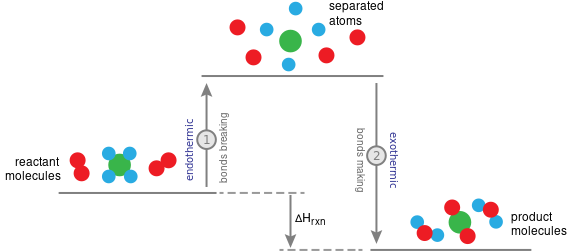
- Lower bond enthalpy → bond breaks more easily.
- Higher bond enthalpy → bond is stronger and harder to break.
Which Bond Breaks First?
- The bond with the lowest bond enthalpy is most likely to break first.
- This is because it requires less energy.
Example
- C–Cl bond (~338 kJ mol\(^{-1}\)) is weaker than C–H (~413 kJ mol\(^{-1}\)).
- Therefore, C–Cl bond breaks more easily.
Effect on Reaction Rate
- Reactions involving weaker bonds occur more easily.
- Lower energy required → lower activation energy.
- Faster reactions at room temperature.
Difficulty of Reaction
- Strong bonds (high bond enthalpy) require more energy to break.
- Reactions are slower or may require heating/catalyst.
Link to Activation Energy
- Bond breaking contributes to activation energy.
- Stronger bonds → higher activation energy → slower reaction.
Important Limitation
- Bond enthalpy alone does not fully determine rate.
- Other factors:
- Activation energy pathway
- Reaction mechanism
- Orientation of molecules
Therefore, bond enthalpy data is useful for predicting reactivity but must be considered alongside other factors.
Example 1:
Predict which bond will break first in the reaction of chloromethane, and explain your answer using bond enthalpy data.
▶️ Answer/Explanation
C–Cl bond has lower bond enthalpy than C–H.
Therefore, C–Cl bond breaks first.
It requires less energy to break.
Example 2:
Explain why iodine reacts more slowly than chlorine in halogenation reactions of alkanes.
▶️ Answer/Explanation
I–I bond is weaker than Cl–Cl, but C–I bond formed is also weaker.
Overall reaction is less energetically favourable.
Therefore, reaction has higher effective activation barrier.
Reaction proceeds more slowly.
