Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 6.2 Exothermic and endothermic reactions-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH22) -Unit 2 – 6.2 Exothermic and endothermic reactions- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH22) -Unit 2 – 6.2 Exothermic and endothermic reactions- Study Notes -International A Level (IAL) Chemistry (YCH22) – per latest Syllabus.
Key Concepts:
6.2 know that, by convention, exothermic reactions have a negative enthalpy change and endothermic reactions have a positive enthalpy change
Edexcel International A Level (IAL) Chemistry (YCH22) -Concise Summary Notes- All Topics
6.2 Sign Convention for Enthalpy Change (ΔH)
By convention, the sign of enthalpy change (\( \Delta H \)) indicates whether heat is released or absorbed during a reaction at constant pressure.
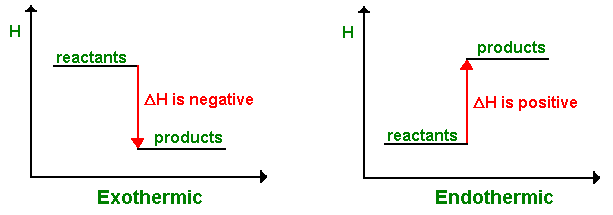
Exothermic Reactions
- Heat is released to the surroundings.
- Surroundings temperature increases.
- Enthalpy change is negative.
\( \Delta H < 0 \)
Explanation
- Energy released when bonds form is greater than energy required to break bonds.
- Excess energy is released as heat.
Example
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
Endothermic Reactions
- Heat is absorbed from the surroundings.
- Surroundings temperature decreases.
- Enthalpy change is positive.
\( \Delta H > 0 \)
Explanation
- More energy is required to break bonds than is released when new bonds form.
- Energy is taken in from surroundings.
Example
\( \mathrm{CaCO_3 \rightarrow CaO + CO_2} \)
Energy Profile Understanding
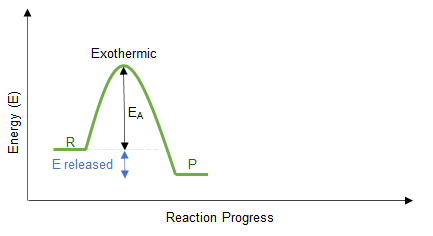
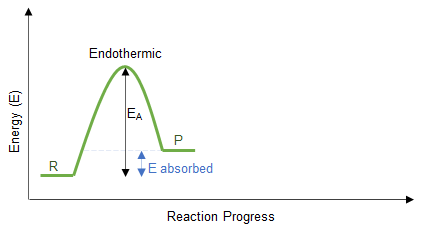
- Exothermic: products have lower energy than reactants.
- Endothermic: products have higher energy than reactants.
Key Points
- \( \Delta H < 0 \) → exothermic reaction.
- \( \Delta H > 0 \) → endothermic reaction.
- Sign convention is based on heat flow between system and surroundings.
Therefore, the sign of \( \Delta H \) provides immediate information about the energy change in a reaction.
Example 1:
A reaction has \( \Delta H = -150 \,\mathrm{kJ\,mol^{-1}} \). Explain what this indicates about the reaction.
▶️ Answer/Explanation
The negative sign indicates an exothermic reaction.
150 kJ mol⁻¹ of heat is released to surroundings.
Therefore, products are lower in energy than reactants.
Example 2:
Explain why bond breaking is endothermic and bond formation is exothermic in terms of enthalpy change.
▶️ Answer/Explanation
Bond breaking requires energy input, so it is endothermic.
Bond formation releases energy, so it is exothermic.
Therefore, overall \( \Delta H \) depends on the balance of these processes.
