Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 6.4 Standard enthalpy changes -Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.4 Standard enthalpy changes – Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.4 Standard enthalpy changes – Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
6.4 know the definition of standard enthalpy change of:
i reaction, ΔrH
ii formation, ΔfH
iii combustion, ΔcH
iv neutralisation, ΔneutH
v atomisation, ΔatH
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
6.4 Standard Enthalpy Changes
Standard enthalpy changes are defined under standard conditions (100 kPa, usually 298 K, substances in standard states). Each type refers to a specific kind of chemical process.
(i) Standard Enthalpy Change of Reaction (ΔrH)
The enthalpy change when the amounts of reactants shown in a balanced equation react completely under standard conditions.
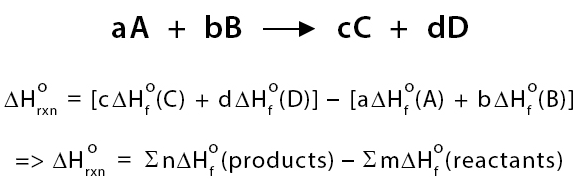
Key Points
- Applies to any reaction.
- Depends on the stoichiometry of the equation.
- Can be exothermic or endothermic.
Example
\( \mathrm{N_2 + 3H_2 \rightarrow 2NH_3} \)
The enthalpy change corresponds exactly to this equation as written.
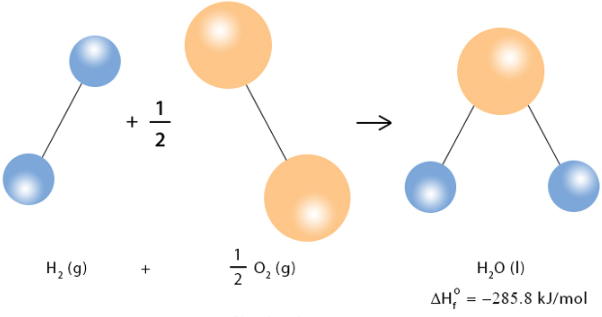
(ii) Standard Enthalpy Change of Formation (ΔfH)
The enthalpy change when one mole of a compound is formed from its constituent elements in their standard states under standard conditions.
Key Points
- Only 1 mole of product is formed.
- Elements must be in their standard states.
- Standard enthalpy of formation of an element = 0.
Example
\( \mathrm{C(graphite) + O_2(g) \rightarrow CO_2(g)} \)
- Forms exactly 1 mole of \( \mathrm{CO_2} \).
- Reactants are in standard states.
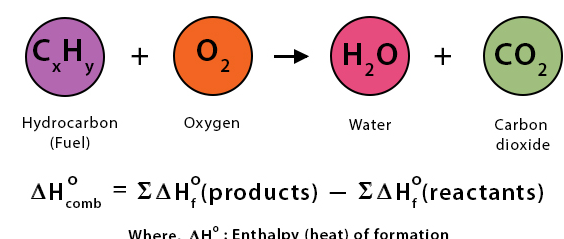
(iii) Standard Enthalpy Change of Combustion (ΔcH)
The enthalpy change when one mole of a substance is completely burned in oxygen under standard conditions.
Key Points
- Oxygen is in excess.
- Complete combustion forms:
- \( \mathrm{CO_2} \) and \( \mathrm{H_2O} \) (for hydrocarbons).
- Always exothermic (\( \Delta H < 0 \)).
Example
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
- 1 mole of methane is burned.
- Complete combustion occurs.
Therefore, each definition depends on specific conditions and the amount of substance involved.
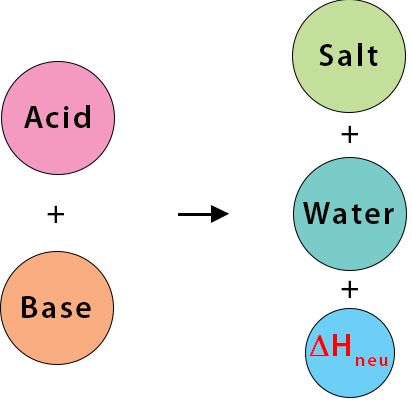
(iv) Standard Enthalpy Change of Neutralisation (ΔneutH)
The enthalpy change when one mole of water is formed by the reaction of an acid with a base under standard conditions.
Key Points
- Always involves formation of 1 mole of \( \mathrm{H_2O} \).
- Typically between acid and alkali.
- Usually exothermic (\( \Delta H < 0 \)).
- For strong acid + strong base, value is approximately constant (~ −57 kJ mol\(^{-1}\)).
Ionic Equation (Key Idea)
\( \mathrm{H^+(aq) + OH^-(aq) \rightarrow H_2O(l)} \)
Example
\( \mathrm{HCl(aq) + NaOH(aq) \rightarrow NaCl(aq) + H_2O(l)} \)
(v) Standard Enthalpy Change of Atomisation (ΔatH)
The enthalpy change when one mole of gaseous atoms is formed from an element in its standard state under standard conditions.

Key Points
- Produces gaseous atoms.
- Always endothermic (\( \Delta H > 0 \)) because bonds are broken.
- Applies to both metals and non-metals.
Examples
\( \mathrm{\frac{1}{2}Cl_2(g) \rightarrow Cl(g)} \)
\( \mathrm{Na(s) \rightarrow Na(g)} \)
Summary
- ΔneutH → formation of 1 mole of water.
- ΔatH → formation of 1 mole of gaseous atoms.
- Neutralisation is exothermic; atomisation is endothermic.
Therefore, these definitions depend on the type of process and the exact quantity formed.
Example 1:
Explain why the following equation is not a standard enthalpy change of formation and correct it:
\( \mathrm{2C(graphite) + O_2 \rightarrow 2CO(g)} \)
▶️ Answer/Explanation
Forms 2 moles of product instead of 1 mole.
Therefore, it does not meet the definition of ΔfH.
Correct equation:
\( \mathrm{C(graphite) + \frac{1}{2}O_2 \rightarrow CO(g)} \)
Example 2:
Distinguish between ΔrH and ΔcH using suitable equations and explain why ΔcH is always negative.
▶️ Answer/Explanation
ΔrH refers to any reaction as written.
Example:
\( \mathrm{N_2 + 3H_2 \rightarrow 2NH_3} \)
ΔcH refers specifically to combustion of 1 mole.
Example:
\( \mathrm{CH_4 + 2O_2 \rightarrow CO_2 + 2H_2O} \)
ΔcH is always negative because combustion releases energy.
Strong bonds form in \( \mathrm{CO_2} \) and \( \mathrm{H_2O} \), releasing large amounts of energy.
Example 3:
Explain why the enthalpy change of neutralisation for a weak acid (e.g. ethanoic acid) is less exothermic than for a strong acid (e.g. HCl).
▶️ Answer/Explanation
Strong acids fully dissociate to give \( \mathrm{H^+} \).
Weak acids only partially dissociate.
Energy is required to ionise the weak acid (endothermic).
Therefore, less heat is released overall.
Example 4:
Explain why the following equation does not represent a standard enthalpy change of atomisation:
\( \mathrm{Cl_2(g) \rightarrow 2Cl(g)} \)
▶️ Answer/Explanation
The equation forms 2 moles of gaseous atoms.
Definition requires formation of 1 mole of gaseous atoms.
Correct equation:
\( \mathrm{\frac{1}{2}Cl_2(g) \rightarrow Cl(g)} \)
