Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 6.5 Calorimetry calculations-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.5 Calorimetry calculations- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.5 Calorimetry calculations- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
6.5 be able to use experimental data to calculate:
i energy transferred in a reaction recalling and using the expression:
energy transferred (J) = mass (g) × specific heat capacity (J g⁻¹ °C⁻¹) × temperature change (°C)
ii enthalpy change of the reaction in kJ mol⁻¹
This will be limited to experiments where substances are mixed in an insulated container and combustion experiments using a suitable calorimeter.
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
6.5 (i) Energy Transferred in a Reaction
In calorimetry experiments, the energy transferred during a reaction is calculated using temperature change of a substance (usually water or solution).
Key Formula
energy transferred (\( q \)) = mass × specific heat capacity × temperature change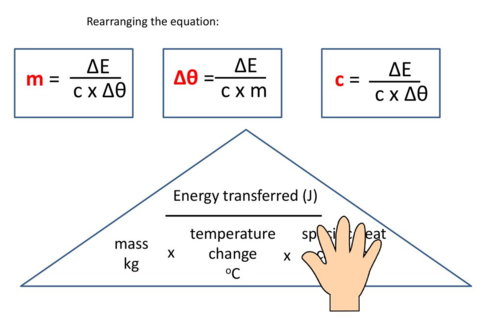
\( q = mc\Delta T \)
Where:
- \( q \) = energy transferred (J)
- \( m \) = mass of solution (g)
- \( c \) = specific heat capacity (J g\(^{-1}\) °C\(^{-1}\))
- \( \Delta T \) = temperature change (°C or K)
Important Notes
- For aqueous solutions, \( c = 4.18 \,\mathrm{J\,g^{-1}\,°C^{-1}} \).
- \( \Delta T = \text{final temperature} – \text{initial temperature} \).
- Assume density of solution ≈ \( 1.00 \,\mathrm{g\,cm^{-3}} \).
Sign of Energy Change
- Temperature increase → exothermic → heat released
- Temperature decrease → endothermic → heat absorbed
Experimental Context
- Reactions carried out in insulated containers (e.g. polystyrene cups).
- Minimises heat loss to surroundings.
- Assumes all heat transfer affects the solution.
Example 1:
50.0 cm³ of solution is heated from 25.0°C to 31.5°C. Calculate the energy transferred.
▶️ Answer/Explanation
Mass = 50.0 g (density ≈ 1.00 g cm\(^{-3}\))
\( \Delta T = 31.5 – 25.0 = 6.5\,°C \)
\( q = mc\Delta T \)
\( q = 50.0 \times 4.18 \times 6.5 \)
\( q = 1358.5 \,\mathrm{J} \approx 1.36 \times 10^3 \,\mathrm{J} \)
Example 2:
In a calorimetry experiment, the temperature of the solution decreases by 4.0°C. Explain what this indicates about the reaction and the sign of \( q \).
▶️ Answer/Explanation
Temperature decrease means heat is absorbed from surroundings.
Reaction is endothermic.
Therefore, \( q \) for the reaction is positive.
6.5 (ii) Enthalpy Change of Reaction (ΔH) in kJ mol\(^{-1}\)
After calculating the energy transferred (\( q \)) using calorimetry, the enthalpy change of the reaction is determined per mole of reactant in kJ mol\(^{-1}\).
Key Steps
- Step 1: Calculate energy transferred using \( q = mc\Delta T \) (in J).
- Step 2: Convert \( q \) to kJ → divide by 1000.
- Step 3: Calculate number of moles of limiting reagent.
- Step 4: Divide energy by moles.
Formula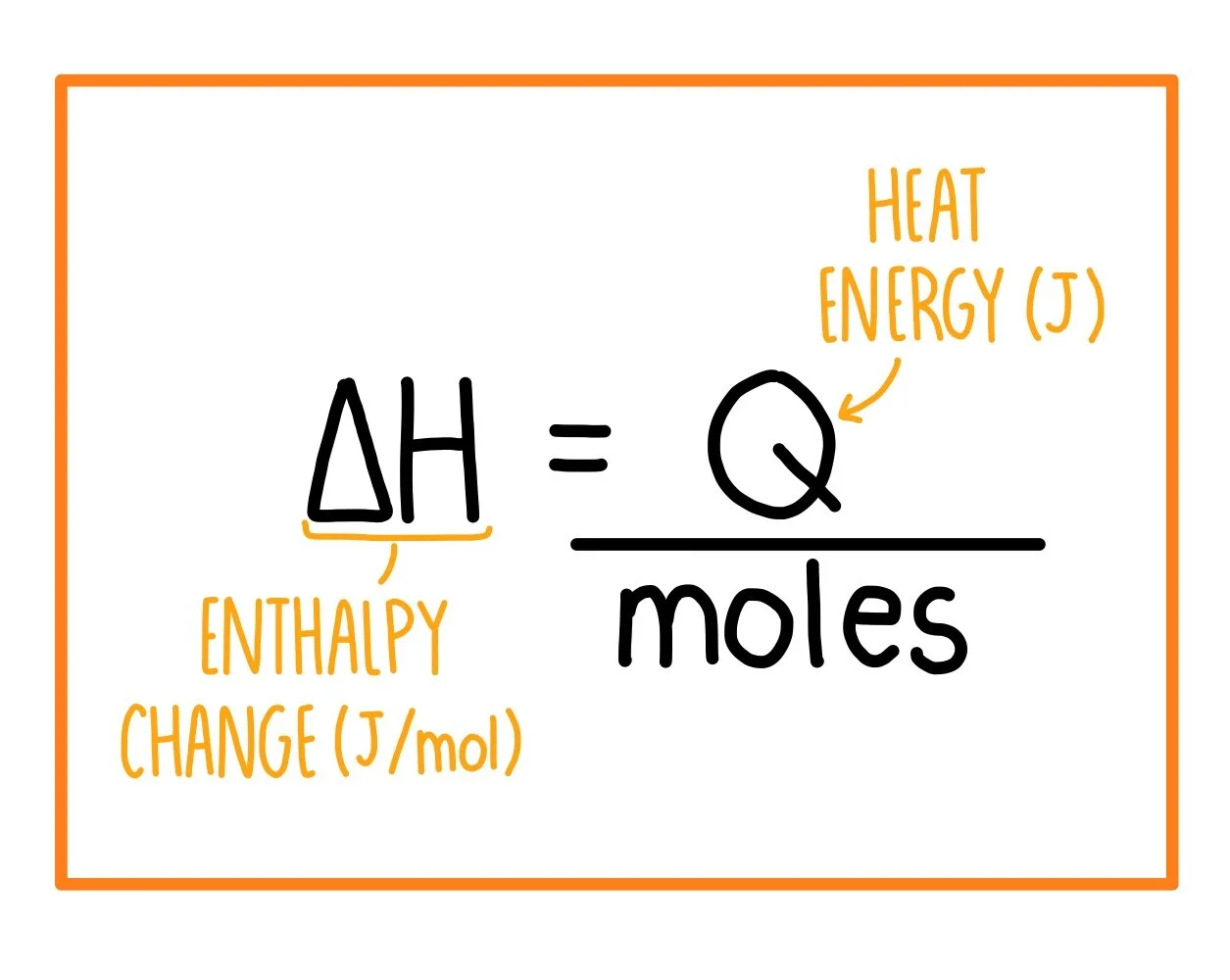
\( \Delta H = \frac{q}{n} \)
(convert \( q \) to kJ first)
Sign Convention
- Temperature increase → exothermic → \( \Delta H < 0 \)
- Temperature decrease → endothermic → \( \Delta H > 0 \)
Important Notes
- Use limiting reagent for moles.
- Units must be kJ mol\(^{-1}\).
- Assume no heat loss (ideal condition).
Example 1:
25.0 cm³ of \( \mathrm{1.00\,mol\,dm^{-3}} \) HCl reacts with 25.0 cm³ of \( \mathrm{1.00\,mol\,dm^{-3}} \) NaOH. Temperature rises by 6.0°C. Calculate ΔH for the reaction.
▶️ Answer/Explanation
Total volume = 50.0 cm³ → mass = 50.0 g
\( q = mc\Delta T \)
\( q = 50.0 \times 4.18 \times 6.0 = 1254 \,\mathrm{J} = 1.254 \,\mathrm{kJ} \)
Moles of HCl = \( \mathrm{1.00 \times \frac{25.0}{1000} = 0.0250\,mol} \)
\( \Delta H = \frac{1.254}{0.0250} = 50.2 \,\mathrm{kJ\,mol^{-1}} \)
Temperature increases → exothermic
Therefore, \( \Delta H = -50.2 \,\mathrm{kJ\,mol^{-1}} \)
Example 2:
A fuel releases 2.50 kJ of energy when 0.0100 mol is burned. Calculate ΔH\(_c\).
▶️ Answer/Explanation
\( \Delta H = \frac{q}{n} \)
\( \Delta H = \frac{2.50}{0.0100} = 250 \,\mathrm{kJ\,mol^{-1}} \)
Combustion releases heat → exothermic
Therefore, \( \Delta H_c = -250 \,\mathrm{kJ\,mol^{-1}} \)
