Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 6.6 Hess’s Law and enthalpy cycles-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.6 Hess’s Law and enthalpy cycles- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.6 Hess’s Law and enthalpy cycles- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
6.6 know Hess’s Law and be able to apply it to:
i constructing enthalpy cycles
ii calculating enthalpy changes of reaction using data provided, or data selected from a table or obtained from experiments
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
6.6 Hess’s Law
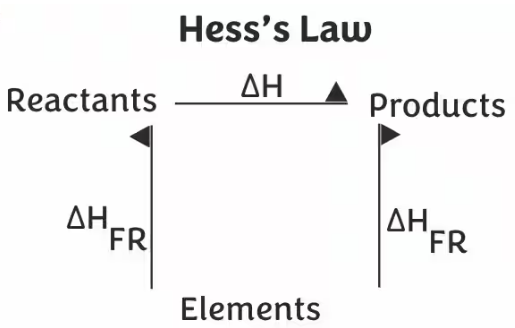
Hess’s Law states that:
The enthalpy change of a reaction is independent of the pathway taken and depends only on the initial and final states.
Key Idea
- Energy is conserved.
- Direct route = indirect route.
- Allows calculation of ΔH when direct measurement is difficult.
(i) Constructing Enthalpy Cycles
An enthalpy cycle is a diagram showing different pathways between reactants and products.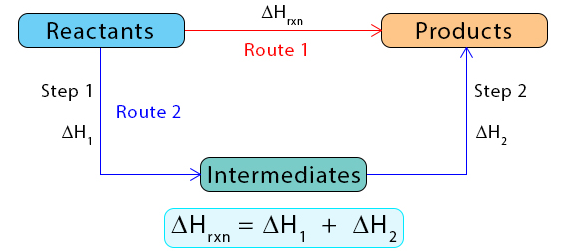
How to Construct a Cycle
- Step 1: Write the target reaction.
- Step 2: Identify known enthalpy changes (e.g. ΔfH or ΔcH).
- Step 3: Draw arrows linking reactants → elements → products (or other route).
- Step 4: Ensure cycle is closed.
Using Formation Enthalpies
\( \Delta H = \sum \Delta_f H (\text{products}) – \sum \Delta_f H (\text{reactants}) \)
Example (Cycle Setup)
\( \mathrm{CO(g) + \frac{1}{2}O_2(g) \rightarrow CO_2(g)} \)
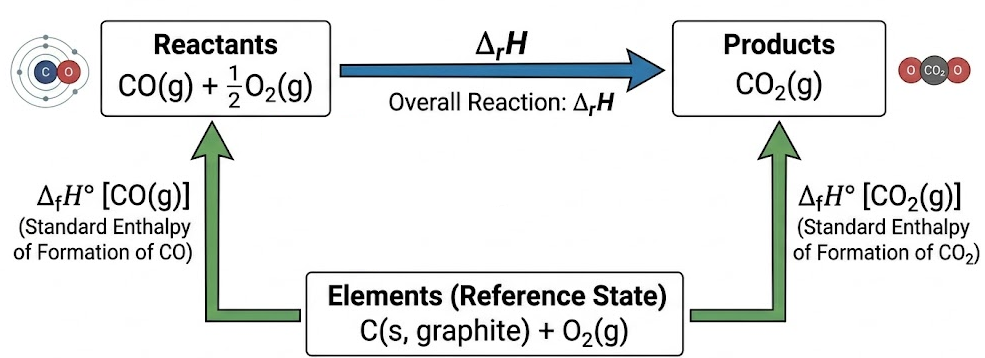
- Use ΔfH values of CO and CO\(_2\).
- Elements (C, O\(_2\)) form intermediate step.
(ii) Calculating Enthalpy Changes Using Hess’s Law
Enthalpy changes can be calculated by:
- Reversing equations → change sign of ΔH
- Multiplying equations → multiply ΔH
- Adding equations → add ΔH values
Example 1 :
Calculate ΔH for:
\( \mathrm{CO(g) + \frac{1}{2}O_2(g) \rightarrow CO_2(g)} \)
Given:
- \( \Delta_f H (\mathrm{CO_2}) = -394 \,\mathrm{kJ\,mol^{-1}} \)
- \( \Delta_f H (\mathrm{CO}) = -111 \,\mathrm{kJ\,mol^{-1}} \)
▶️ Answer/Explanation
\( \Delta H = \sum \Delta_f H (\text{products}) – \sum \Delta_f H (\text{reactants}) \)
\( \Delta H = (-394) – (-111) \)
\( \Delta H = -283 \,\mathrm{kJ\,mol^{-1}} \)
Example 2:
Find ΔH for:
\( \mathrm{C(graphite) + \frac{1}{2}O_2(g) \rightarrow CO(g)} \)
Given:
\( \mathrm{C + O_2 \rightarrow CO_2} \quad \Delta H = -394 \,\mathrm{kJ\,mol^{-1}} \)
\( \mathrm{CO + \frac{1}{2}O_2 \rightarrow CO_2} \quad \Delta H = -283 \,\mathrm{kJ\,mol^{-1}} \)
▶️ Answer/Explanation
Reverse second equation:
\( \mathrm{CO_2 \rightarrow CO + \frac{1}{2}O_2} \quad \Delta H = +283 \)
Add equations:
\( \mathrm{C + O_2 \rightarrow CO_2} \)
\( \mathrm{CO_2 \rightarrow CO + \frac{1}{2}O_2} \)
Overall:
\( \mathrm{C + \frac{1}{2}O_2 \rightarrow CO} \)
\( \Delta H = -394 + 283 = -111 \,\mathrm{kJ\,mol^{-1}} \)
