Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 6.7 CORE PRACTICAL 2: Hess’s Law-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.7 CORE PRACTICAL 2: Hess’s Law- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.7 CORE PRACTICAL 2: Hess’s Law- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
6.7 CORE PRACTICAL 2
Determination of the enthalpy change of a reaction using Hess’s Law
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
6.7 CORE PRACTICAL 2: Determination of Enthalpy Change using Hess’s Law
This core practical determines the enthalpy change of a reaction that cannot be measured directly, by using Hess’s Law and experimental data from related reactions.
Aim
To determine the enthalpy change of a reaction using Hess’s Law by measuring enthalpy changes of two related reactions.
Typical Reactions Used
Example system (commonly used):
Reaction 1: \( \mathrm{NaOH(aq) + HCl(aq) \rightarrow NaCl(aq) + H_2O(l)} \)
Reaction 2: \( \mathrm{Na_2CO_3(aq) + 2HCl(aq) \rightarrow 2NaCl(aq) + H_2O(l) + CO_2(g)} \)
From these, ΔH for another reaction (e.g. involving \( \mathrm{NaHCO_3} \)) can be calculated using Hess’s Law.
Apparatus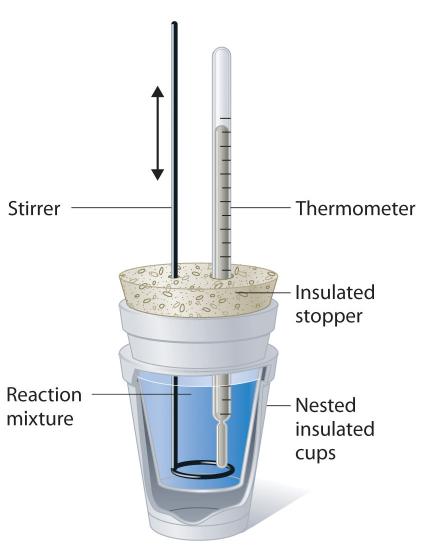
- Polystyrene cup (insulated calorimeter)
- Thermometer
- Measuring cylinder / pipette
- Stirrer
Procedure (General)
- Measure known volumes of reactants.
- Record initial temperature.
- Mix reactants in insulated cup.
- Stir and record maximum/minimum temperature.
- Repeat for second reaction.
Calculations
Step 1: Calculate energy change
\( q = mc\Delta T \)
Step 2: Calculate ΔH for each reaction
\( \Delta H = \frac{q}{n} \) (convert to kJ mol\(^{-1}\))
Step 3: Apply Hess’s Law
- Add/subtract equations appropriately.
- Combine ΔH values.
Key Assumptions
- No heat loss to surroundings.
- Density = \( \mathrm{1.00\,g\,cm^{-3}} \).
- Specific heat capacity = \( \mathrm{4.18\,J\,g^{-1}\,°C^{-1}} \).
Sources of Error
- Heat loss to surroundings.
- Incomplete mixing.
- Inaccurate temperature readings.
Improvements
- Use lid on calorimeter.
- Stir continuously.
- Use more precise thermometer or data logger.
Conclusion
- ΔH for reactions can be determined indirectly.
- Hess’s Law ensures validity of calculation.
Therefore, this practical demonstrates how experimental data and theory combine to determine enthalpy changes.
Example 1:
Two enthalpy changes are measured:
Reaction A: \( \Delta H = -57 \,\mathrm{kJ\,mol^{-1}} \)
Reaction B: \( \Delta H = -100 \,\mathrm{kJ\,mol^{-1}} \)
If the target reaction = A − B, calculate ΔH.
▶️ Answer/Explanation
\( \Delta H = -57 – (-100) \)
\( \Delta H = +43 \,\mathrm{kJ\,mol^{-1}} \)
Example 2:
Explain why Hess’s Law is necessary in this practical instead of measuring the enthalpy change directly.
▶️ Answer/Explanation
Some reactions are too slow or difficult to measure directly.
Others may involve side reactions or experimental limitations.
Hess’s Law allows calculation using measurable reactions.
Therefore, indirect determination is more practical and reliable.
