Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 6.9–6.10 Bond enthalpy and calculations-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.9–6.10 Bond enthalpy and calculations- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 6.9–6.10 Bond enthalpy and calculations- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
6.9 understand the terms ‘bond enthalpy’ and ‘mean bond enthalpy’, and be able to use bond enthalpies to calculate enthalpy changes, understanding the limitations of this method
6.10 be able to calculate mean bond enthalpies from enthalpy changes of reaction
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
6.9 Bond Enthalpy and Mean Bond Enthalpy
Bond Enthalpy
The bond enthalpy is the energy required to break one mole of a specific covalent bond in the gaseous state.
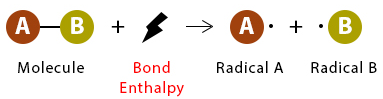
- Always endothermic (\( \Delta H > 0 \)).
- Depends on the specific molecule.
Example
\( \mathrm{H_2(g) \rightarrow 2H(g)} \)
Mean Bond Enthalpy
The mean bond enthalpy is the average energy required to break a particular type of bond in different molecules.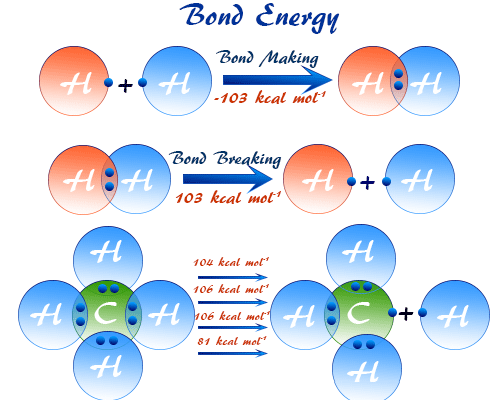
- Averaged over many compounds.
- Used when exact bond enthalpy is not available.
Example
- C–H bond enthalpy varies slightly in different molecules.
- Mean value is used for calculations.
Using Bond Enthalpies to Calculate ΔH
Key Formula
\( \Delta H = \sum \text{(bond enthalpies of bonds broken)} – \sum \text{(bond enthalpies of bonds formed)} \)
Steps
- Step 1: Draw full structural formula.
- Step 2: Identify bonds broken (reactants).
- Step 3: Identify bonds formed (products).
- Step 4: Apply formula.
Therefore, bond enthalpies provide a useful but approximate way to estimate enthalpy changes.
Example 1:
Calculate ΔH for:
\( \mathrm{H_2 + Cl_2 \rightarrow 2HCl} \)
Given bond enthalpies (kJ mol\(^{-1}\)):
- H–H = 436
- Cl–Cl = 242
- H–Cl = 431
▶️ Answer/Explanation
Bonds broken:
H–H + Cl–Cl = \( 436 + 242 = 678 \)
Bonds formed:
2(H–Cl) = \( 2 \times 431 = 862 \)
\( \Delta H = 678 – 862 = -184 \,\mathrm{kJ\,mol^{-1}} \)
Example 2:
Estimate ΔH for:
\( \mathrm{CH_4 + Cl_2 \rightarrow CH_3Cl + HCl} \)
(Use appropriate bond enthalpy values.)
▶️ Answer/Explanation
Bonds broken: 1 C–H and 1 Cl–Cl
Bonds formed: 1 C–Cl and 1 H–Cl
Substitute values and calculate using formula.
Final ΔH is slightly exothermic.
6.10 Calculating Mean Bond Enthalpies from Enthalpy Changes
Mean bond enthalpies can be determined using experimental enthalpy change data and the bond enthalpy equation.
Key Idea
\( \Delta H = \sum \text{(bond enthalpies of bonds broken)} – \sum \text{(bond enthalpies of bonds formed)} \)
If one bond enthalpy is unknown, it can be calculated by rearranging this equation.
Method
- Step 1: Write balanced equation.
- Step 2: Identify all bonds broken and formed.
- Step 3: Substitute known bond enthalpies and ΔH.
- Step 4: Rearrange to find unknown bond enthalpy.
Limitations
- Values are mean (average), not exact.
- Depends on molecular environment.
- Only applies to gaseous species.
Summary
- Use \( \Delta H = \text{broken} – \text{formed} \).
- Rearrange to find unknown bond enthalpy.
- Method gives approximate values.
Therefore, experimental ΔH data can be used to estimate mean bond enthalpies.
Example 1 :
The reaction has \( \Delta H = -184 \,\mathrm{kJ\,mol^{-1}} \):
\( \mathrm{H_2 + Cl_2 \rightarrow 2HCl} \)
Given:
- H–H = 436
- Cl–Cl = 242
- H–Cl = ?
▶️ Answer/Explanation
Bonds broken = \( 436 + 242 = 678 \)
Bonds formed = \( 2 \times \text{(H–Cl)} \)
\( -184 = 678 – 2x \)
\( 2x = 678 + 184 = 862 \)
\( x = 431 \,\mathrm{kJ\,mol^{-1}} \)
Example 2 :
The enthalpy change for the reaction is \( \Delta H = -74 \,\mathrm{kJ\,mol^{-1}} \):
\( \mathrm{CH_4 + Cl_2 \rightarrow CH_3Cl + HCl} \)
Given:
- C–H = 413
- Cl–Cl = 242
- H–Cl = 431
- C–Cl = ?
▶️ Answer/Explanation
Bonds broken = \( 413 + 242 = 655 \)
Bonds formed = \( x + 431 \)
\( -74 = 655 – (x + 431) \)
\( -74 = 224 – x \)
\( x = 298 \,\mathrm{kJ\,mol^{-1}} \)
