Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 7.1 Types of intermolecular forces-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH22) -Unit 2 – 7.1 Types of intermolecular forces- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.1 Types of intermolecular forces- Study Notes -International A Level (IAL) Chemistry (YCH22) – per latest Syllabus.
Key Concepts:
7.1 understand the nature of the following intermolecular forces:
i London forces (instantaneous dipole-induced dipole)
ii permanent dipole–permanent dipole interactions
iii hydrogen bonds
Edexcel International A Level (IAL) Chemistry (YCH22) -Concise Summary Notes- All Topics
7.1 (i) London Forces (Instantaneous Dipole–Induced Dipole)
London forces are the weakest type of intermolecular force and are present in all molecules, especially in non-polar substances.
Definition
London forces arise from temporary fluctuations in electron distribution, which create instantaneous dipoles that induce dipoles in neighbouring molecules.
How They Arise (Step-by-Step)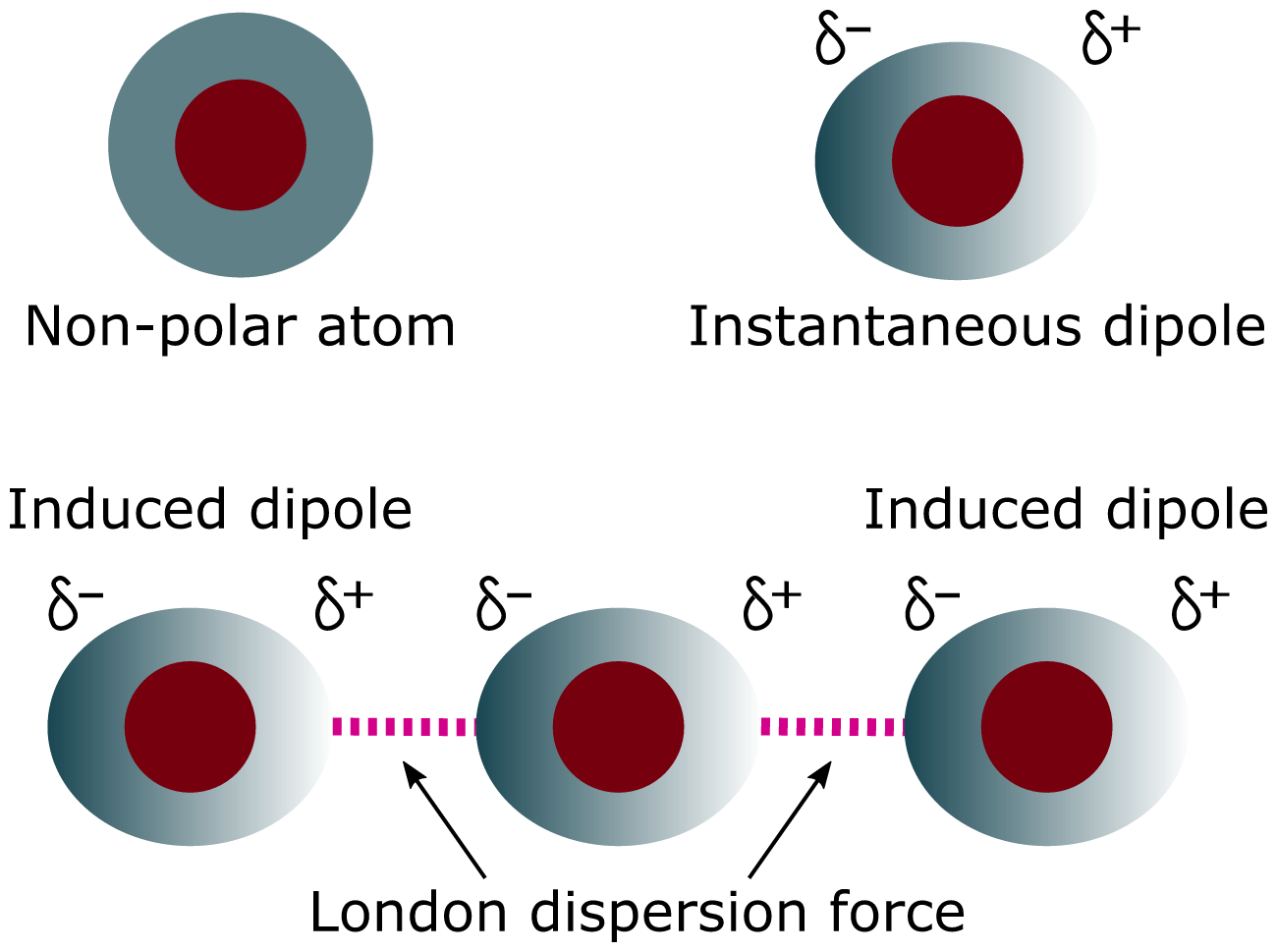
- Electrons are constantly moving.
- At any instant, electrons may be unevenly distributed.
- This creates an instantaneous dipole.
- This dipole induces a dipole in a neighbouring molecule.
- Weak electrostatic attraction forms between them.
Key Features
- Present in all molecules.
- Only force in non-polar molecules.
- Very weak compared to other intermolecular forces.
- Strength increases with:
- Number of electrons
- Size of molecule (molar mass)
- Surface area (long chains > compact molecules)
Examples
- Noble gases: \( \mathrm{He, Ne, Ar} \)
- Non-polar molecules: \( \mathrm{CH_4, Cl_2} \)
Effect on Physical Properties
- Stronger London forces → higher boiling point.
- Larger molecules have stronger forces → higher melting/boiling points.
Trend Example
- \( \mathrm{CH_4 < C_2H_6 < C_3H_8} \) (increasing boiling point)
- Due to increasing number of electrons → stronger London forces.
Therefore, London forces explain why even non-polar substances can exist as liquids or solids.
Example 1 :
Explain why iodine (\( \mathrm{I_2} \)) has a much higher boiling point than chlorine (\( \mathrm{Cl_2} \)).
▶️ Answer/Explanation
\( \mathrm{I_2} \) has more electrons than \( \mathrm{Cl_2} \).
Stronger London forces are formed.
More energy required to overcome these forces.
Therefore, higher boiling point.
Example 2 :
Explain why straight-chain alkanes have higher boiling points than branched isomers.
▶️ Answer/Explanation
Straight-chain molecules have larger surface area.
More contact between molecules → stronger London forces.
Branched molecules are more compact → weaker forces.
Therefore, lower boiling point for branched isomers.
7.1 (ii) Permanent Dipole–Permanent Dipole Interactions
Permanent dipole–dipole interactions occur between polar molecules that have a permanent separation of charge.
Definition
These are electrostatic attractions between the δ⁺ (partial positive) end of one molecule and the δ⁻ (partial negative) end of another molecule.
How They Arise
- Occur in molecules with polar bonds.
- Due to difference in electronegativity between atoms.
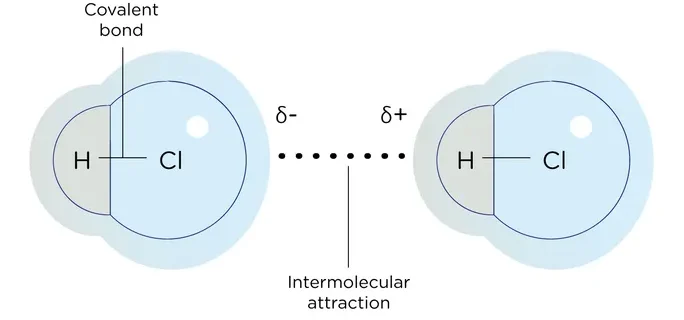
- If molecule is asymmetrical → permanent dipole forms.
Example
- \( \mathrm{HCl} \):
- H = δ⁺, Cl = δ⁻
- Molecules attract via dipole–dipole forces.
Key Features
- Occur only in polar molecules.
- Stronger than London forces (for similar size molecules).
- Weaker than hydrogen bonding.
Conditions for Occurrence
- Molecule must have:
- Polar bonds
- Asymmetrical shape (net dipole)
Effect on Physical Properties
- Stronger intermolecular forces → higher boiling point.
- Polar molecules generally have higher boiling points than non-polar molecules of similar size.
Comparison Example
- \( \mathrm{HCl} \) vs \( \mathrm{Cl_2} \)
- \( \mathrm{HCl} \): dipole–dipole + London forces
- \( \mathrm{Cl_2} \): only London forces
- Therefore, \( \mathrm{HCl} \) has stronger intermolecular forces.
Therefore, permanent dipole–dipole interactions explain why polar molecules have stronger intermolecular attractions than non-polar ones.
Example 1:
Explain why \( \mathrm{HCl} \) has a higher boiling point than \( \mathrm{Cl_2} \), even though they have similar molar masses.
▶️ Answer/Explanation
\( \mathrm{HCl} \) is polar and has permanent dipole–dipole interactions.
\( \mathrm{Cl_2} \) is non-polar and only has London forces.
Dipole–dipole forces are stronger.
Therefore, \( \mathrm{HCl} \) has a higher boiling point.
Example 2:
Explain why \( \mathrm{CO_2} \) does not exhibit permanent dipole–dipole interactions, despite having polar bonds.
▶️ Answer/Explanation
\( \mathrm{CO_2} \) has polar C=O bonds.
Molecule is linear and symmetrical.
Dipoles cancel out.
Therefore, no permanent dipole exists.
7.1 (iii) Hydrogen Bonding
Hydrogen bonding is a strong type of intermolecular force that occurs in molecules containing hydrogen bonded to highly electronegative atoms.
Definition
A hydrogen bond is the strong electrostatic attraction between a hydrogen atom (δ⁺), covalently bonded to a highly electronegative atom (N, O, or F), and a lone pair of electrons on another N, O, or F atom.
Conditions for Hydrogen Bonding
- Hydrogen must be bonded to:
- \( \mathrm{N} \), \( \mathrm{O} \), or \( \mathrm{F} \)
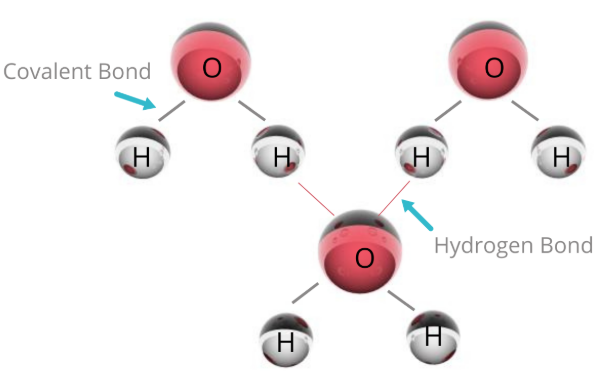
- \( \mathrm{N} \), \( \mathrm{O} \), or \( \mathrm{F} \)
- Presence of lone pair on neighbouring molecule
Examples
- Water: \( \mathrm{H_2O} \)
- Ammonia: \( \mathrm{NH_3} \)
- Hydrogen fluoride: \( \mathrm{HF} \)
How It Arises
- N, O, F are highly electronegative.
- Bond with H becomes highly polar.
- H becomes strongly δ⁺.
- Strong attraction forms with lone pair on nearby molecule.
Key Features
- Stronger than dipole–dipole interactions.
- Still weaker than covalent bonds.
- Directional (specific orientation required).
Effect on Physical Properties
- Much higher boiling points.
- Increased viscosity.
- Greater solubility in water.
Important Example
- \( \mathrm{H_2O} \) has unusually high boiling point.
- Due to extensive hydrogen bonding network.
Comparison with Other Forces
- London forces < Dipole–dipole < Hydrogen bonding
Therefore, hydrogen bonding plays a crucial role in determining the physical properties of many important substances.
Example 1 :
Explain why water has a much higher boiling point than methane (\( \mathrm{CH_4} \)).
▶️ Answer/Explanation
Water forms hydrogen bonds between molecules.
Methane only has London forces.
Hydrogen bonds are much stronger.
Therefore, more energy is required to separate water molecules.
Example 2:
Explain why hydrogen chloride (\( \mathrm{HCl} \)) does not form hydrogen bonds.
▶️ Answer/Explanation
Hydrogen bonding requires H bonded to N, O, or F.
Chlorine is less electronegative than these elements.
Therefore, HCl does not form strong hydrogen bonds.
