Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 7.2 Hydrogen bonding-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.2 Hydrogen bonding- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.2 Hydrogen bonding- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
7.2 understand the interactions in molecules, such as H₂O, liquid NH₃ and liquid HF, which give rise to hydrogen bonding
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
7.2 Hydrogen Bonding in Specific Molecules
Hydrogen bonding arises in molecules such as \( \mathrm{H_2O} \), \( \mathrm{NH_3} \), and \( \mathrm{HF} \) due to the presence of highly polar bonds and lone pairs on electronegative atoms.
General Requirement Recap
- H bonded to \( \mathrm{N} \), \( \mathrm{O} \), or \( \mathrm{F} \)
- Lone pair on neighbouring molecule
(i) Water (\( \mathrm{H_2O} \))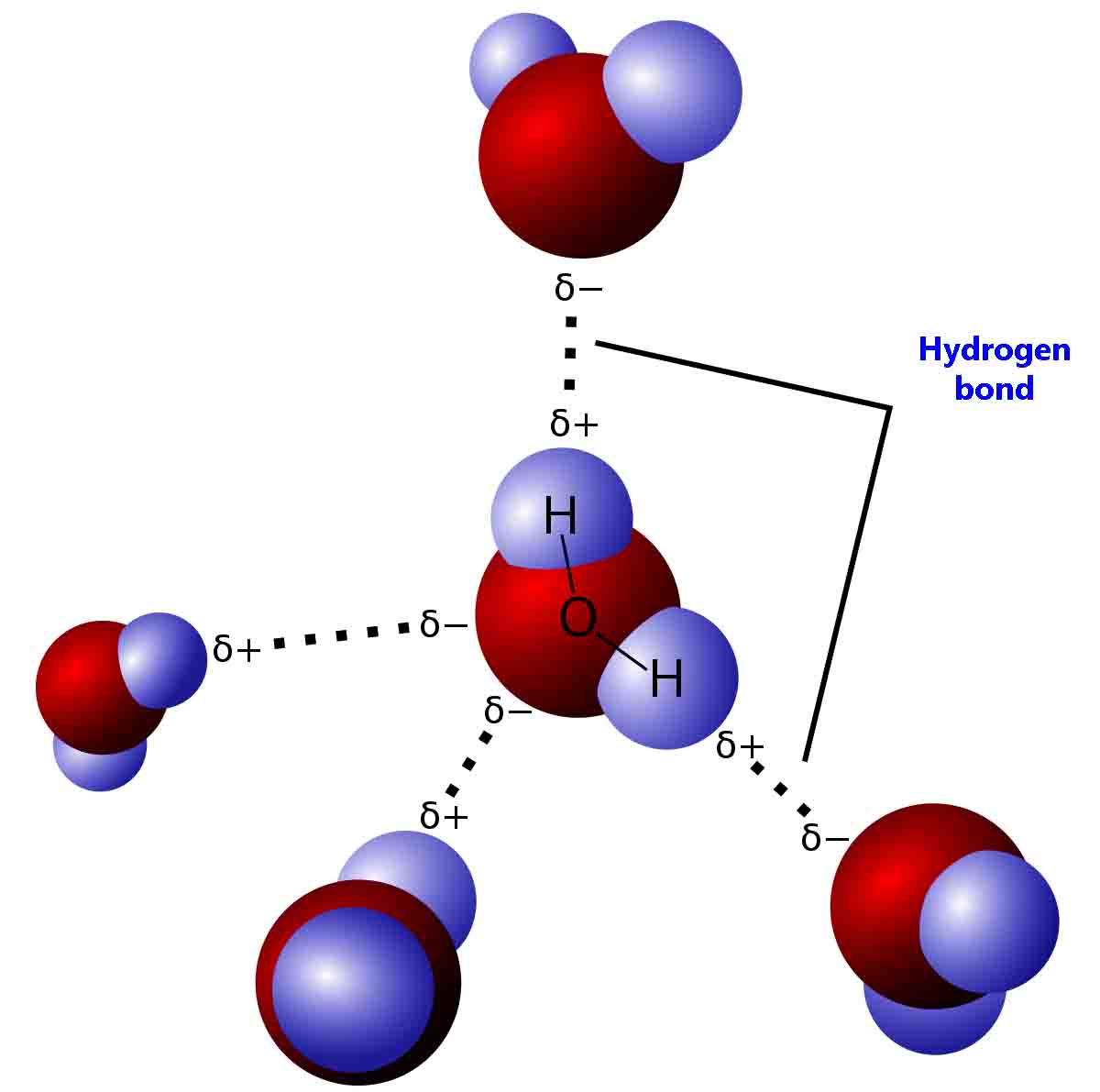
Structure and Bonding
- O–H bonds are highly polar.
- Oxygen has 2 lone pairs.
Hydrogen Bonding Pattern
- Each molecule can form up to 4 hydrogen bonds:
- 2 via hydrogen atoms
- 2 via lone pairs on oxygen
Result
- Extensive 3D hydrogen-bonded network.
- Very high boiling point for small molecule.
(ii) Ammonia (\( \mathrm{NH_3} \))
Structure and Bonding
- N–H bonds are polar.
- Nitrogen has 1 lone pair.
Hydrogen Bonding Pattern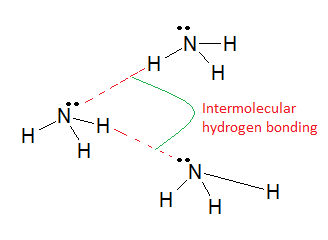
- Each molecule can form fewer hydrogen bonds than water:
- 3 H atoms available
- Only 1 lone pair available
Result
- Less extensive hydrogen bonding network than water.
- Lower boiling point than water.
(iii) Hydrogen Fluoride (\( \mathrm{HF} \))
Structure and Bonding
- H–F bond is very polar.
- Fluorine has 3 lone pairs.
Hydrogen Bonding Pattern
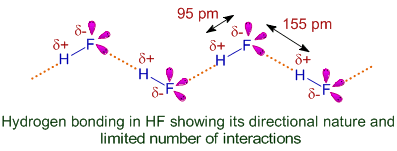
- Forms chains of molecules via hydrogen bonding.
- Each molecule typically forms 2 hydrogen bonds.
Result
- Strong hydrogen bonding due to very high electronegativity of F.
- Higher boiling point than expected for its size.
Comparison Summary
- \( \mathrm{H_2O} \): extensive 3D network → strongest overall effect
- \( \mathrm{NH_3} \): fewer H-bonds → weaker network
- \( \mathrm{HF} \): strong but forms chains, not 3D network
Key Insight
- Strength of hydrogen bonding depends on:
- Electronegativity (F > O > N)
- Number of hydrogen bonds formed
- Structure (network vs chain)
The nature and extent of hydrogen bonding depend on both electronegativity and molecular structure.
Example 1 :
Explain why water has a higher boiling point than ammonia despite ammonia having hydrogen bonding.
▶️ Answer/Explanation
Water forms more hydrogen bonds per molecule (up to 4).
Ammonia forms fewer hydrogen bonds.
Water has a stronger, more extensive network.
Therefore, more energy is needed to separate molecules.
Example 2 :
Explain why HF has a higher boiling point than HCl.
▶️ Answer/Explanation
HF forms hydrogen bonds due to H–F bond.
HCl only has dipole–dipole interactions.
Hydrogen bonding is much stronger.
Therefore, HF has a higher boiling point.
