Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 7.4 Predicting hydrogen bonding-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.4 Predicting hydrogen bonding- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.4 Predicting hydrogen bonding- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
7.4 be able to predict the presence of hydrogen bonding in molecules analogous to those mentioned in 7.2
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
7.4 Predicting Hydrogen Bonding in Molecules
To predict whether a molecule shows hydrogen bonding, you must check for specific structural features similar to \( \mathrm{H_2O} \), \( \mathrm{NH_3} \), and \( \mathrm{HF} \).
Essential Conditions
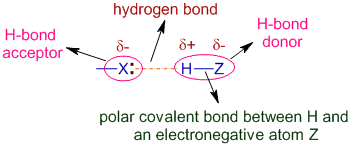
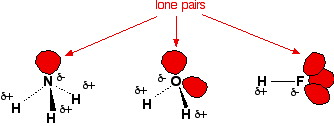
- Hydrogen must be directly bonded to:
- \( \mathrm{N} \), \( \mathrm{O} \), or \( \mathrm{F} \)
- A neighbouring molecule must have:
- A lone pair on \( \mathrm{N} \), \( \mathrm{O} \), or \( \mathrm{F} \)
Prediction Rule
If a molecule contains H–N, H–O, or H–F bonds, it can form hydrogen bonds.
Examples That DO Show Hydrogen Bonding
- \( \mathrm{CH_3OH} \) (methanol) → contains O–H
- \( \mathrm{C_2H_5OH} \) (ethanol) → contains O–H
- \( \mathrm{CH_3NH_2} \) (methylamine) → contains N–H
- \( \mathrm{HF} \) → contains H–F
Examples That DO NOT Show Hydrogen Bonding
- \( \mathrm{CH_4} \) → no N, O, F
- \( \mathrm{HCl} \) → H–Cl not strong enough
- \( \mathrm{CH_3OCH_3} \) → has O but no O–H bond
Important Insight
- Having N, O, or F alone is not enough.
- Must have H directly bonded to them.
Intermolecular vs Intramolecular
- Usually intermolecular (between molecules).
- Occasionally intramolecular (within same molecule).
Effect on Properties
- Higher boiling points.
- Greater solubility in water.
Step-by-Step Prediction Method
- Step 1: Look for N, O, or F.
- Step 2: Check if H is bonded to them.
- Step 3: Confirm presence of lone pairs.
- Step 4: Conclude hydrogen bonding possible.
Therefore, hydrogen bonding can be predicted by identifying specific bonding patterns in molecules.
Example 1:
Which of the following molecules can form hydrogen bonds: \( \mathrm{CH_3OH} \), \( \mathrm{CH_3Cl} \), \( \mathrm{NH_3} \)? Explain.
▶️ Answer/Explanation
\( \mathrm{CH_3OH} \): yes (O–H bond).
\( \mathrm{NH_3} \): yes (N–H bond).
\( \mathrm{CH_3Cl} \): no (no H–N/O/F bond).
Example 2:
Explain why ethanol has a higher boiling point than dimethyl ether (\( \mathrm{CH_3OCH_3} \)).
▶️ Answer/Explanation
Ethanol has O–H bonds → hydrogen bonding.
Dimethyl ether lacks O–H bonds.
Only weaker intermolecular forces present.
Therefore, ethanol has stronger intermolecular forces and higher boiling point.
