Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 7.5 IMF and physical properties-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.5 IMF and physical properties- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.5 IMF and physical properties- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
7.5 understand, in terms of intermolecular forces, physical properties shown by substances, including:
i the trends in boiling temperatures of alkanes with increasing chain length
ii the effect of branching in the carbon chain on the boiling temperatures of alkanes
iii the relatively low volatility (higher boiling temperatures) of alcohols compared to alkanes with a similar number of electrons
iv the trends in boiling temperatures of the hydrogen halides HF to HI
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
7.5 Intermolecular Forces and Boiling Temperature
(i) Trend in Boiling Temperatures of Alkanes with Increasing Chain Length
Observation
- Boiling temperature increases as carbon chain length increases.
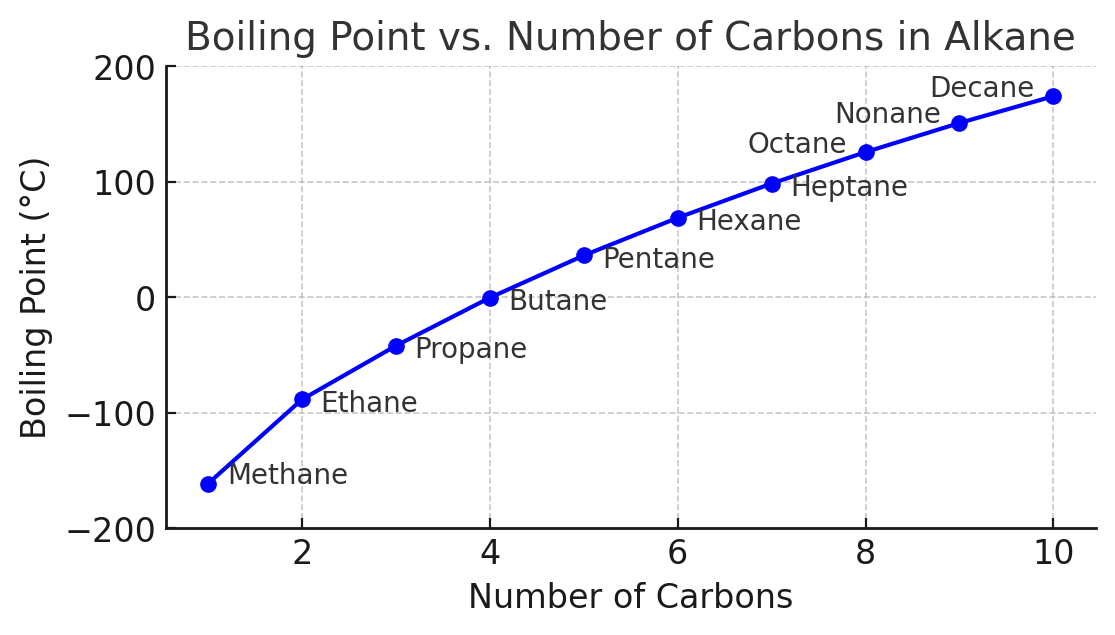
- Example:
- \( \mathrm{CH_4 < C_2H_6 < C_3H_8 < C_4H_{10}} \)
Explanation
- Alkanes are non-polar molecules.
- Only intermolecular forces present are London dispersion forces.
- As chain length increases:
- Number of electrons increases
- Molecular size increases
- Surface area increases
- This leads to:
- Stronger temporary dipoles
- Stronger London forces
- More energy required to separate molecules → higher boiling temperature.
Key Conclusion
Increasing chain length → stronger intermolecular forces → higher boiling temperature.
(ii) Effect of Branching on Boiling Temperatures of Alkanes
Observation
- Increased branching → lower boiling temperature.
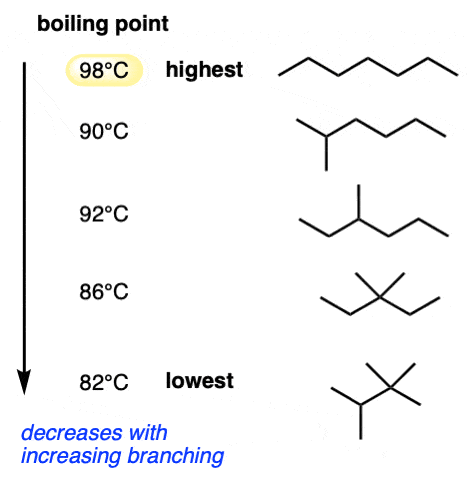
- Example:
- \( \mathrm{C_5H_{12}} \):
- Pentane (straight chain) → highest b.p.
- 2-methylbutane → lower b.p.
- 2,2-dimethylpropane → lowest b.p.
Explanation
- Branching makes molecules more compact and spherical.
- This reduces surface area of contact between molecules.
- As a result:
- London dispersion forces become weaker
- Less energy required to separate molecules
- Therefore, boiling temperature decreases.
Conclusion
More branching → smaller surface area → weaker intermolecular forces → lower boiling temperature.
Summary
- Longer chains → stronger London forces → higher boiling point.
- More branching → weaker London forces → lower boiling point.
Therefore, boiling temperature in alkanes depends on molecular size and shape.
Example 1:
Explain why \( \mathrm{C_6H_{14}} \) has a higher boiling temperature than \( \mathrm{C_4H_{10}} \).
▶️ Answer/Explanation
\( \mathrm{C_6H_{14}} \) has more electrons and larger molecular size.
Stronger London dispersion forces are present.
More energy required to separate molecules.
Therefore, higher boiling temperature.
Example 2:
Explain why 2,2-dimethylpropane has a lower boiling temperature than pentane.
▶️ Answer/Explanation
2,2-dimethylpropane is highly branched.
Molecule is more compact with smaller surface area.
Weaker London dispersion forces.
Therefore, lower boiling temperature.
(iii) Higher Boiling Temperatures of Alcohols vs Alkanes
Observation
- Alcohols have higher boiling temperatures than alkanes with similar number of electrons.
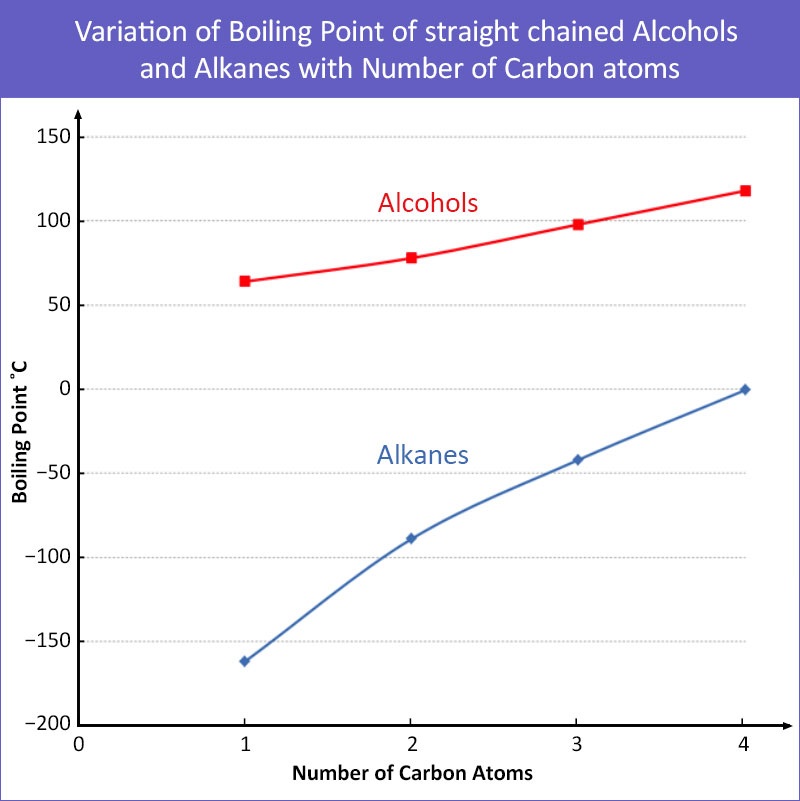
- Example:
- \( \mathrm{C_2H_5OH} \) vs \( \mathrm{C_2H_6} \)
Explanation
- Alkanes: only London dispersion forces.
- Alcohols:
- London dispersion forces
- Permanent dipole–dipole interactions
- Hydrogen bonding (strongest)
- Hydrogen bonding occurs due to:
- Highly polar O–H bond
- Attraction between \( \mathrm{H^{\delta+}} \) and lone pair on O
- Stronger intermolecular forces → more energy needed to break → higher boiling temperature.
Conclusion
Presence of hydrogen bonding in alcohols significantly increases boiling temperature compared to alkanes.
(iv) Trends in Boiling Temperatures of Hydrogen Halides (HF → HI)
Observation
- General trend (HCl → HI): boiling temperature increases down the group.
- However, HF has an anomalously high boiling temperature.
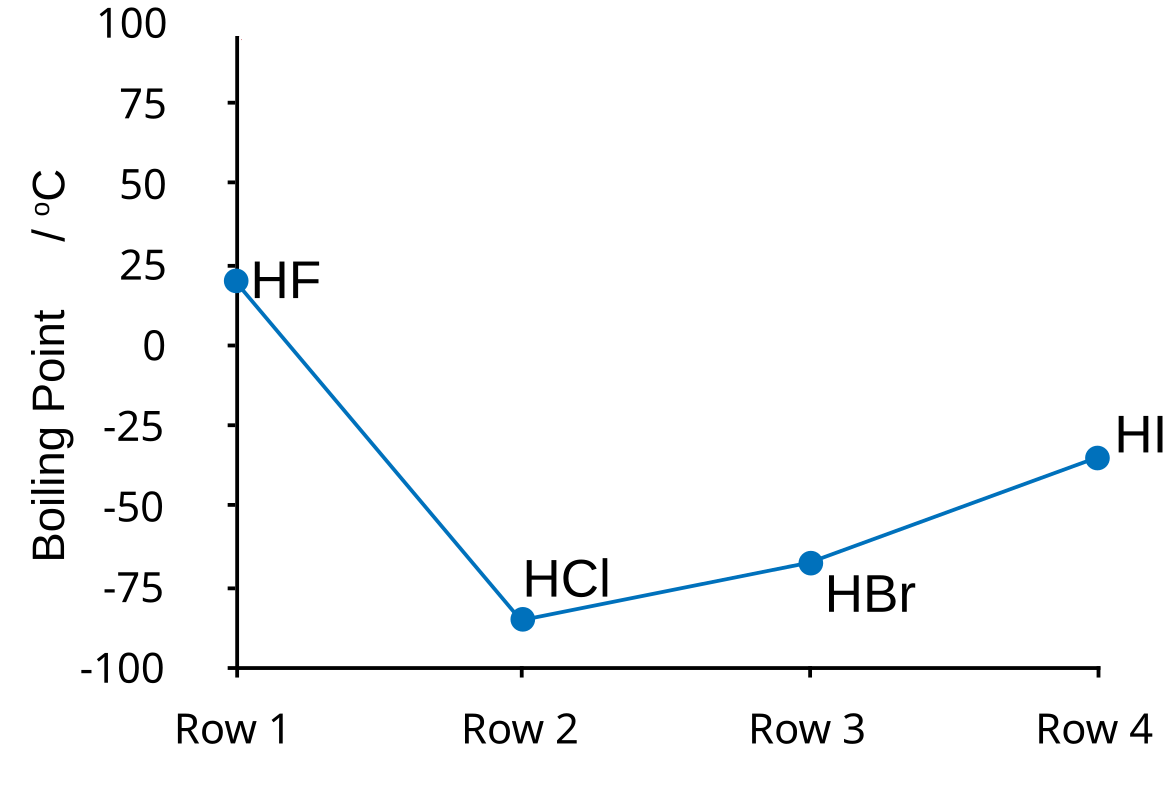
From HCl to HI:
- Increasing number of electrons.
- Stronger London dispersion forces.
- Therefore, boiling temperature increases.
Why HF is Anomalous
- HF molecules form hydrogen bonds.
- Fluorine is highly electronegative.
- Strong intermolecular attraction between molecules.
- Therefore:
- HF has much higher boiling temperature than expected.
Trend Summary
\( \mathrm{HF > HI > HBr > HCl} \) (in terms of boiling temperature)
Conclusion
- HF: hydrogen bonding dominates.
- HCl → HI: London forces dominate.
Summary
- Alcohols have higher boiling temperatures due to hydrogen bonding.
- Hydrogen halides show increasing boiling temperature down the group due to London forces.
- HF is anomalous due to hydrogen bonding.
Therefore, boiling temperature depends on the type and strength of intermolecular forces.
Example 1 :
Explain why ethanol has a higher boiling temperature than ethane.
▶️ Answer/Explanation
Ethanol forms hydrogen bonds due to O–H group.
Ethane only has London dispersion forces.
Hydrogen bonds are stronger.
Therefore, more energy required to separate ethanol molecules.
Example 2:
Explain the trend in boiling temperatures of hydrogen halides from HCl to HI and why HF deviates.
▶️ Answer/Explanation
From HCl to HI, number of electrons increases.
Stronger London dispersion forces.
Therefore, boiling temperature increases.
HF forms hydrogen bonds due to highly polar H–F bond.
This leads to unusually high boiling temperature.
