Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 7.6 Solvent choice and intermolecular forces-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.6 Solvent choice and intermolecular forces- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 7.6 Solvent choice and intermolecular forces- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
7.6 understand factors that influence the choice of solvents, including:
i water, to dissolve some ionic compounds, in terms of the hydration of the ions
ii water, to dissolve simple alcohols, in terms of hydrogen bonding
iii water, as a poor solvent for compounds (to include polar molecules such as halogenoalkane), in terms of inability to form hydrogen bonds
iv non-aqueous solvents, for compounds that have similar intermolecular forces to those in the solvent
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
7.6 Factors Influencing Choice of Solvents
(i) Water as a Solvent for Ionic Compounds
Key Idea: Hydration of Ions
Water dissolves ionic compounds because ions become surrounded by water molecules (hydration).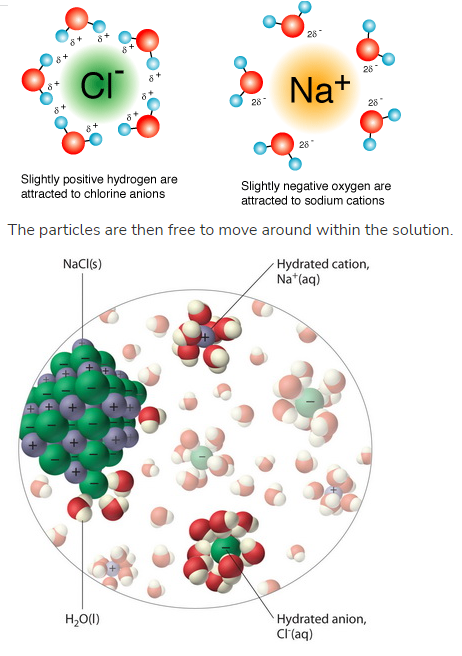
Explanation
- Water is a polar molecule:
- Oxygen has \( \delta^- \) charge
- Hydrogen has \( \delta^+ \) charge
- When an ionic compound dissolves:
- Positive ions attract the \( \delta^- \) oxygen end
- Negative ions attract the \( \delta^+ \) hydrogen end
- This forms ion–dipole interactions.
- Ions become hydrated and separated from the lattice.
Example
\( \mathrm{NaCl(s) \rightarrow Na^+(aq) + Cl^-(aq)} \)
Key Conclusion
Water dissolves ionic compounds due to strong ion–dipole attractions (hydration).
(ii) Water as a Solvent for Simple Alcohols
Key Idea: Hydrogen Bonding
Simple alcohols dissolve in water because they can form hydrogen bonds with water molecules.
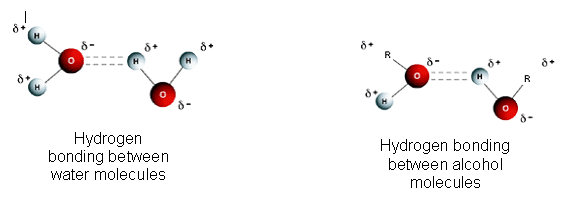
Explanation
- Alcohols contain –OH group.
- This group is polar.
- Hydrogen bonding occurs between:
- \( \mathrm{H^{\delta+}} \) of –OH
- Lone pair on oxygen of water
- These strong interactions allow alcohol molecules to mix with water.
Effect of Chain Length
- Small alcohols (e.g. methanol, ethanol) → fully miscible.
- Larger alcohols → less soluble.
- Due to increasing non-polar hydrocarbon chain.
Example
\( \mathrm{C_2H_5OH(l) + H_2O(l)} \) → hydrogen bonding between molecules
Key Conclusion
Alcohols dissolve in water due to hydrogen bonding between –OH groups and water molecules.
Summary
- Ionic compounds dissolve due to hydration (ion–dipole forces).
- Alcohols dissolve due to hydrogen bonding.
Therefore, solubility in water depends on the ability to form strong interactions with water molecules.
Example 1:
Explain why sodium chloride dissolves readily in water but not in hexane.
▶️ Answer/Explanation
Water is polar and forms ion–dipole interactions with ions.
Hexane is non-polar and cannot stabilise ions.
Therefore, NaCl dissolves in water but not in hexane.
Example 2 :
Explain why ethanol is soluble in water but hexanol is only slightly soluble.
▶️ Answer/Explanation
Both form hydrogen bonds via –OH group.
Hexanol has a longer non-polar hydrocarbon chain.
Non-polar part reduces overall polarity.
Therefore, solubility decreases with chain length.
(iii) Water as a Poor Solvent for Some Compounds
Key Idea: Inability to Form Hydrogen Bonds
Water is a poor solvent for many organic compounds because they cannot form hydrogen bonds with water.
Explanation
- Water is polar and forms strong hydrogen bonds.
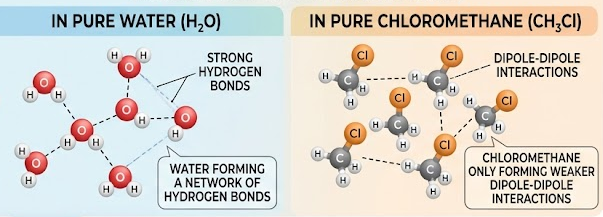
- Some compounds (e.g. halogenoalkanes):
- May be slightly polar
- But do not form hydrogen bonds with water
- Therefore:
- Weak interactions with water
- Cannot replace strong water–water hydrogen bonds
- Result: substance is insoluble or only slightly soluble.
Example
Halogenoalkanes (e.g. \( \mathrm{CH_3Cl} \)) are not soluble in water.
Conclusion
If a substance cannot form hydrogen bonds with water, it is unlikely to dissolve.
(iv) Non-aqueous Solvents and “Like Dissolves Like”
A substance dissolves best in a solvent that has similar intermolecular forces.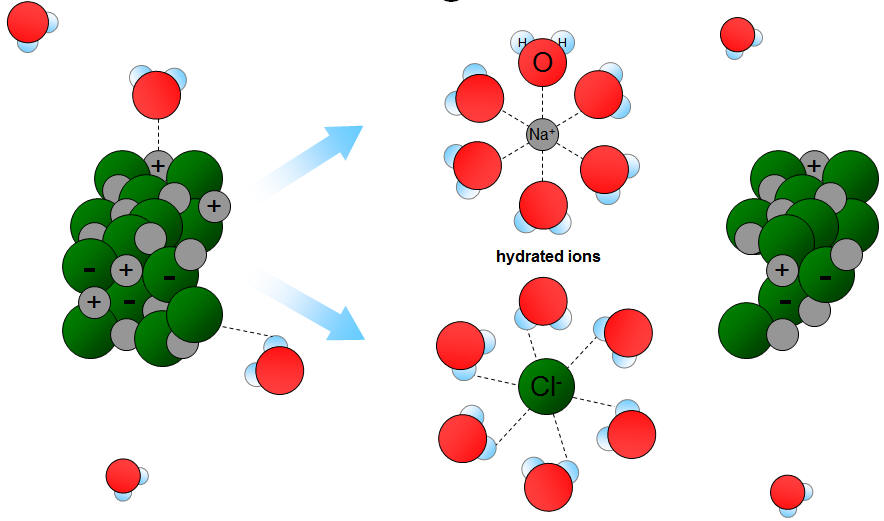
Explanation
- Non-aqueous solvents (e.g. hexane) are non-polar.
- They interact via London dispersion forces.
- Non-polar solutes:
- Also have London forces
- Therefore dissolve easily
- Polar solutes:
- Do not dissolve well in non-polar solvents
Examples
- Iodine dissolves in hexane (both non-polar).
- Oil dissolves in organic solvents, not in water.
Summary
- Water cannot dissolve substances that cannot hydrogen bond.
- Solubility depends on similarity of intermolecular forces.
- “Like dissolves like” explains solvent choice.
Therefore, solvent selection depends on intermolecular interactions between solute and solvent.
Example 1 :
Explain why chloromethane is only slightly soluble in water.
▶️ Answer/Explanation
Chloromethane is polar but cannot form hydrogen bonds with water.
Water–water hydrogen bonds are strong.
Weak interactions cannot replace them.
Therefore, low solubility.
Example 2 :
Explain why iodine dissolves in hexane but not in water.
▶️ Answer/Explanation
Iodine is non-polar.
Hexane is non-polar → similar London forces.
Therefore, iodine dissolves in hexane.
Water is polar and cannot interact effectively.
Therefore, iodine is insoluble in water.
