Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.10 Ionic half-equations-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.10 Ionic half-equations- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.10 Ionic half-equations- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.10 be able to write ionic half-equations and use them to construct full ionic equations
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.10 Ionic Half-Equations and Full Ionic Equations
Ionic half-equations are used to represent oxidation and reduction processes separately. These can then be combined to form a full ionic equation for a redox reaction.
What is a Half-Equation?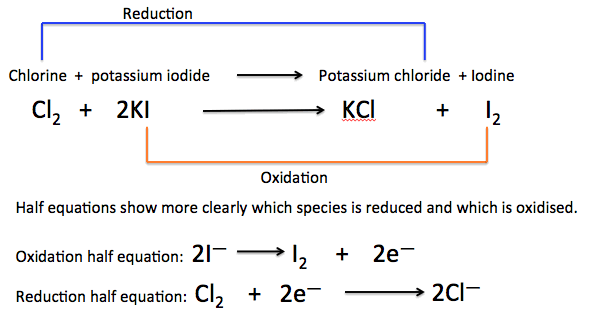
A half-equation shows either loss or gain of electrons.
- Oxidation half-equation → electrons are lost
- Reduction half-equation → electrons are gained
Example of Half-Equations
- Oxidation: \( \mathrm{Zn \rightarrow Zn^{2+} + 2e^-} \)
- Reduction: \( \mathrm{Cu^{2+} + 2e^- \rightarrow Cu} \)
Steps to Construct Full Ionic Equation
- Step 1: Write oxidation and reduction half-equations.
- Step 2: Balance electrons (same number lost and gained).
- Step 3: Add the half-equations together.
- Step 4: Cancel electrons to obtain full ionic equation.
Half-equations:
Balancing Half-Equations (Important Rules)
- Balance atoms first.
- Balance charge using electrons.
- In acidic conditions, use \( \mathrm{H^+} \) and \( \mathrm{H_2O} \).
Example (Acidic Conditions)
Reduction of manganate(VII):
\( \mathrm{MnO_4^- + 8H^+ + 5e^- \rightarrow Mn^{2+} + 4H_2O} \)
Exam Points
- Oxidation = loss of electrons.
- Reduction = gain of electrons.
- Electrons must cancel when combining equations.
- Charges must balance on both sides.
Therefore, half-equations provide a systematic way to construct balanced redox equations.
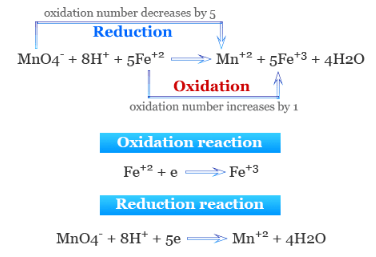
Example 1 :
Iron(II) ions are oxidised by acidified manganate(VII) ions.
(a) Write the half-equations for both processes.
(b) Construct the overall ionic equation.
▶️ Answer/Explanation
(a)
Oxidation: \( \mathrm{Fe^{2+} \rightarrow Fe^{3+} + e^-} \)
Reduction: \( \mathrm{MnO_4^- + 8H^+ + 5e^- \rightarrow Mn^{2+} + 4H_2O} \)
(b)
Multiply iron equation by 5:
\( \mathrm{5Fe^{2+} \rightarrow 5Fe^{3+} + 5e^-} \)
Add both equations:
\( \mathrm{MnO_4^- + 8H^+ + 5Fe^{2+} \rightarrow Mn^{2+} + 4H_2O + 5Fe^{3+}} \)
Example 2:
Chlorine reacts with iodide ions to form iodine and chloride ions.
(a) Write the half-equations.
(b) Deduce the full ionic equation.
▶️ Answer/Explanation
(a)
Oxidation: \( \mathrm{2I^- \rightarrow I_2 + 2e^-} \)
Reduction: \( \mathrm{Cl_2 + 2e^- \rightarrow 2Cl^-} \)
(b)
Add equations:
\( \mathrm{Cl_2 + 2I^- \rightarrow 2Cl^- + I_2} \)
Electrons cancel correctly.
