Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.11 Ionisation energy trends-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.11 Ionisation energy trends- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.11 Ionisation energy trends- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.11 understand reasons for the trend in ionisation energy down Groups 1 and 2
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.11 Ionisation Energy Trend in Group 1
First Ionisation Energy
The energy required to remove one electron from each atom in one mole of gaseous atoms to form one mole of gaseous 1+ ions.
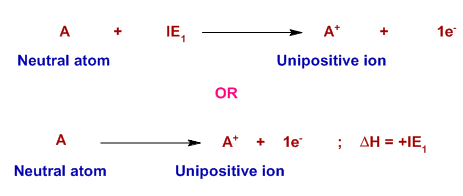
\( \mathrm{X(g) \rightarrow X^+(g) + e^-} \)
Trend in Group 1
- First ionisation energy decreases down Group 1.
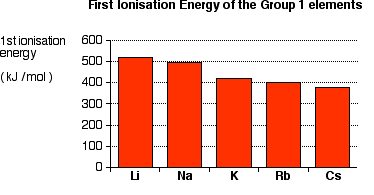
\( \mathrm{Li > Na > K > Rb > Cs} \)
Reason 1: Increasing Atomic Radius
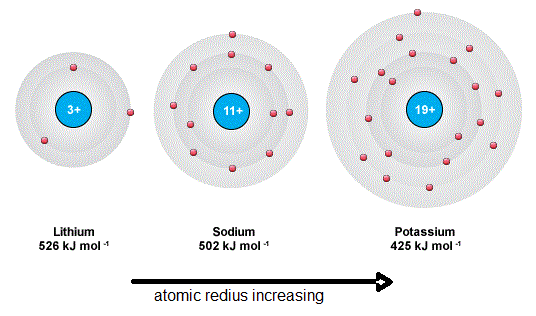
- Each element has an extra electron shell.
- Outer electron is further from the nucleus.
- Weaker electrostatic attraction.
Reason 2: Increased Shielding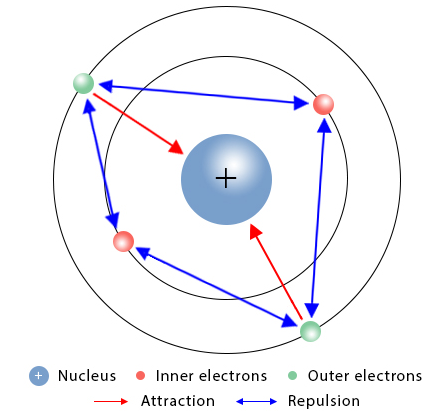
- More inner electron shells.
- Inner electrons shield outer electron from nucleus.
- Reduces effective nuclear attraction.
Reason 3: Nuclear Charge Effect is Overpowered
- Proton number increases down the group.
- BUT increased shielding and distance have a greater effect.
Overall Explanation
As you go down Group 1, the outer electron is further from the nucleus and more shielded by inner shells. This reduces the attraction between the nucleus and the outer electron, so less energy is required to remove it.
Example:
Explain why sodium has a lower first ionisation energy than lithium.
▶️ Answer/Explanation
Sodium has one more electron shell than lithium.
The outer electron is further from the nucleus.
There is greater shielding by inner electrons.
Therefore, the attraction between the nucleus and outer electron is weaker, so less energy is required to remove it.
8.11 Ionisation Energy Trend in Group 2
Second Ionisation Energy
The energy required to remove one electron from each ion in one mole of gaseous 1+ ions to form one mole of gaseous 2+ ions.
\( \mathrm{X^+(g) \rightarrow X^{2+}(g) + e^-} \)
Trend in Group 2
- First ionisation energy decreases down Group 2.
\( \mathrm{Be > Mg > Ca > Sr > Ba} \)
Reason 1: Increasing Atomic Radius
- Each element has an additional electron shell.
- Outer electron is further from the nucleus.
- Attraction between nucleus and electron decreases.
Reason 2: Increased Shielding
- More inner shells → more shielding.
- Reduces effective nuclear attraction on outer electron.
Reason 3: Nuclear Charge Increase is Outweighed
- Number of protons increases down the group.
- BUT shielding + distance increase more significantly.
Key Difference vs Group 1
- Group 2 atoms have two outer electrons.
- Slightly higher ionisation energies than Group 1 in same period.
- Due to higher nuclear charge with similar shielding.
Overall Explanation
Ionisation energy decreases down Group 2 because the outer electron is further from the nucleus and increasingly shielded by inner shells. Although nuclear charge increases, this is outweighed by increased distance and shielding, so less energy is required to remove the electron.
Example :
Explain why calcium has a lower first ionisation energy than magnesium.
▶️ Answer/Explanation
Calcium has an extra electron shell compared to magnesium.
The outer electron is further from the nucleus.
There is greater shielding by inner electrons.
Therefore, the attraction between nucleus and outer electron is weaker, so less energy is required to remove it.
