Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.12 Reactivity trends-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.12 Reactivity trends- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.12 Reactivity trends- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.12 understand reasons for the trend in reactivity of the elements down Group 1 (Li to K) and Group 2 (Mg to Ba)
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.12 Trends in Reactivity Down Group 1 and Group 2
The reactivity of metals in Group 1 and Group 2 increases down the group. This is explained by changes in atomic structure and the ease of electron loss.
Reactivity of metals depends on how easily they lose electrons to form positive ions.
Group 1 (Li → Na → K)
General Trend
- Reactivity increases down the group.
Explanation
- Atomic radius increases.
- Outer electron is further from nucleus.
- Increased shielding effect from inner shells.
- Weaker attraction between nucleus and outer electron.
- Ionisation energy decreases.
- Electron is lost more easily → reactivity increases.
Example Reaction (with water)
\( \mathrm{2Na + 2H_2O \rightarrow 2NaOH + H_2} \)
- Reaction becomes more vigorous from Li → K.
Group 2 (Mg → Ca → Sr → Ba)
General Trend
- Reactivity also increases down the group.
Explanation
- Metals lose two electrons to form \( \mathrm{M^{2+}} \).
- Atomic radius increases down the group.
- Shielding increases.
- Attraction between nucleus and outer electrons decreases.
- Ionisation energies decrease.
- Easier to lose electrons → higher reactivity.
Example Reaction (with water)
\( \mathrm{Ca + 2H_2O \rightarrow Ca(OH)_2 + H_2} \)
- Mg reacts slowly, Ca reacts more readily, Ba reacts vigorously.
Comparison: Group 1 vs Group 2
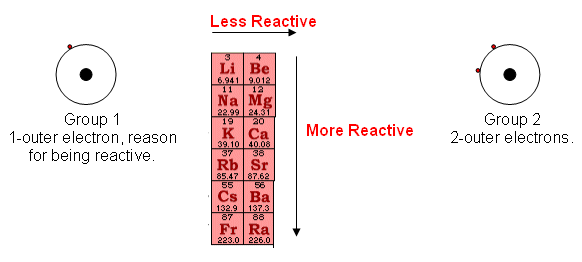
- Group 1 metals are more reactive than Group 2.
- Reason:
- Group 1 loses 1 electron.
- Group 2 loses 2 electrons (requires more energy).
Key Factors Summary
- Increasing atomic radius ↓
- Increasing shielding ↓
- Decreasing ionisation energy ↓
- → Easier electron loss → Increased reactivity
Therefore, the trend in reactivity is controlled by how easily atoms can lose their outer electrons.
Example 1 :
Explain why potassium reacts more vigorously with water than lithium.
▶️ Answer/Explanation
Potassium has a larger atomic radius.
More shielding reduces nuclear attraction.
Outer electron is lost more easily.
Therefore, higher reactivity.
Example 2:
Explain why magnesium is less reactive than calcium.
▶️ Answer/Explanation
Calcium has a larger atomic radius than magnesium.
More shielding reduces attraction to outer electrons.
Electrons are lost more easily in calcium.
Therefore, calcium is more reactive.
