Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.13 Reactions with O₂, Cl₂ and H₂O-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.13 Reactions with O₂, Cl₂ and H₂O- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.13 Reactions with O₂, Cl₂ and H₂O- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.13 know the reactions of the elements of Group 1 (Li to K) and Group 2 (Mg to Ba) with oxygen, chlorine and water
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.13 Reactions of Group 1 and Group 2 Elements
Group 1 and Group 2 metals react with oxygen, chlorine, and water to form characteristic compounds. Reactivity generally increases down the group.
(A) Group 1 (Li → Na → K)
(i) Reaction with Oxygen
Group 1 metals form oxides, peroxides, or superoxides depending on the metal.

- \( \mathrm{4Li + O_2 \rightarrow 2Li_2O} \) (oxide)
- \( \mathrm{2Na + O_2 \rightarrow Na_2O_2} \) (peroxide)
- \( \mathrm{K + O_2 \rightarrow KO_2} \) (superoxide)
Reactivity increases down group.
(ii) Reaction with Chlorine
All Group 1 metals react vigorously with chlorine to form ionic chlorides.
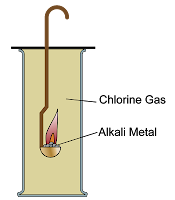
\( \mathrm{2Na + Cl_2 \rightarrow 2NaCl} \)
White crystalline solids formed.
(iii) Reaction with Water
Group 1 metals react vigorously with water to form alkaline hydroxides and hydrogen gas.
\( \mathrm{2K + 2H_2O \rightarrow 2KOH + H_2} \)
- Reactivity increases Li → K.
- Potassium may ignite (lilac flame).
(B) Group 2 (Mg → Ca → Sr → Ba)
(i) Reaction with Oxygen
Group 2 metals form oxides.
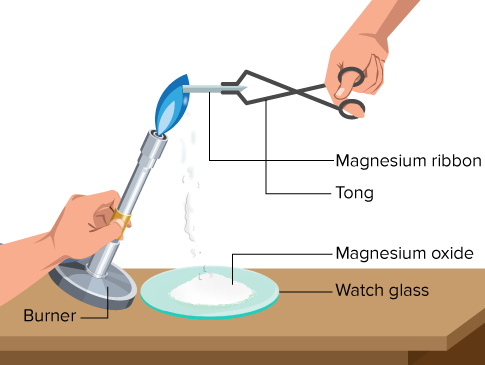
\( \mathrm{2Mg + O_2 \rightarrow 2MgO} \)
Magnesium burns with bright white flame.
(ii) Reaction with Chlorine
Group 2 metals react with chlorine to form MCl\(_2\).
\( \mathrm{Mg + Cl_2 \rightarrow MgCl_2} \)
Ionic compounds formed.
(iii) Reaction with Water
Magnesium
- Reacts slowly with cold water:

\( \mathrm{Mg + 2H_2O \rightarrow Mg(OH)_2 + H_2} \)
- Reacts faster with steam:
\( \mathrm{Mg + H_2O(g) \rightarrow MgO + H_2} \)
Calcium, Strontium, Barium
- React readily with cold water.
\( \mathrm{Ca + 2H_2O \rightarrow Ca(OH)_2 + H_2} \)
- Reactivity increases down the group.
Comparison
- Both groups form oxides and chlorides.
- Both react with water to produce hydroxides + hydrogen.
- Group 1 reacts more vigorously than Group 2.
Trend
- Reactivity increases down both groups.
- Due to decreasing ionisation energy.
Summary
- Group 1: forms oxides/peroxides/superoxides, reacts vigorously with water.
- Group 2: forms oxides, reacts less vigorously with water (except heavier metals).
Therefore, both groups show similar reaction types but differ in intensity and products.
Example 1:
Explain why potassium forms a superoxide while lithium forms an oxide when reacting with oxygen.
▶️ Answer/Explanation
Larger K\(^+\) ion stabilises larger superoxide ion.
Smaller Li\(^+\) ion stabilises smaller oxide ion.
Therefore, different products form.
Example 2 :
Explain why magnesium reacts slowly with cold water but calcium reacts readily.
▶️ Answer/Explanation
Calcium has lower ionisation energy than magnesium.
Electrons are lost more easily.
Therefore, calcium reacts more readily.