Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.16 Thermal stability trends-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.16 Thermal stability trends- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.16 Thermal stability trends- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.16 understand the reasons for the trends in thermal stability of the nitrates and the carbonates of the elements in Groups 1 and 2 in terms of the size and charge of the cations involved
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.16 Thermal Stability of Group 1 and Group 2 Nitrates and Carbonates
Thermal stability refers to how easily a compound decomposes when heated. For nitrates and carbonates of Group 1 and Group 2, thermal stability increases down the group.
Thermal stability depends on the polarising power of the cation.
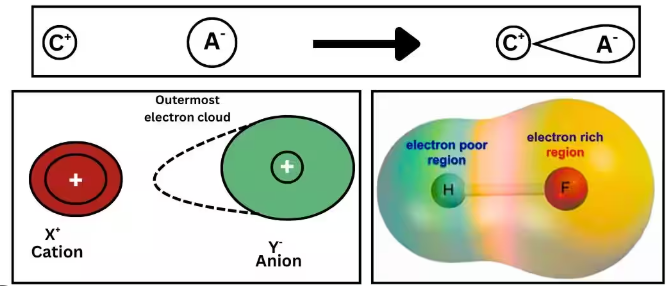
Polarising Power
- Small, highly charged cations have high charge density.
- They distort (polarise) the electron cloud of the anion.
- This weakens bonds in the anion → easier decomposition.
Trend Down the Group
- Cation size increases.
- Charge density decreases.
- Polarising power decreases.
- Anion is less distorted.
- Compound becomes more thermally stable.
(A) Carbonates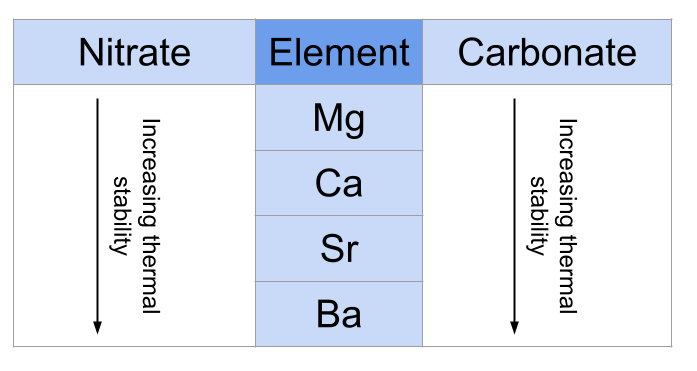
General Decomposition
\( \mathrm{MCO_3 \rightarrow MO + CO_2} \)
Trend
- Stability increases down both groups.
- More heat required for decomposition.
Example
- \( \mathrm{MgCO_3} \) decomposes easily.
- \( \mathrm{BaCO_3} \) is more stable.
(B) Nitrates
Group 1 (except Li)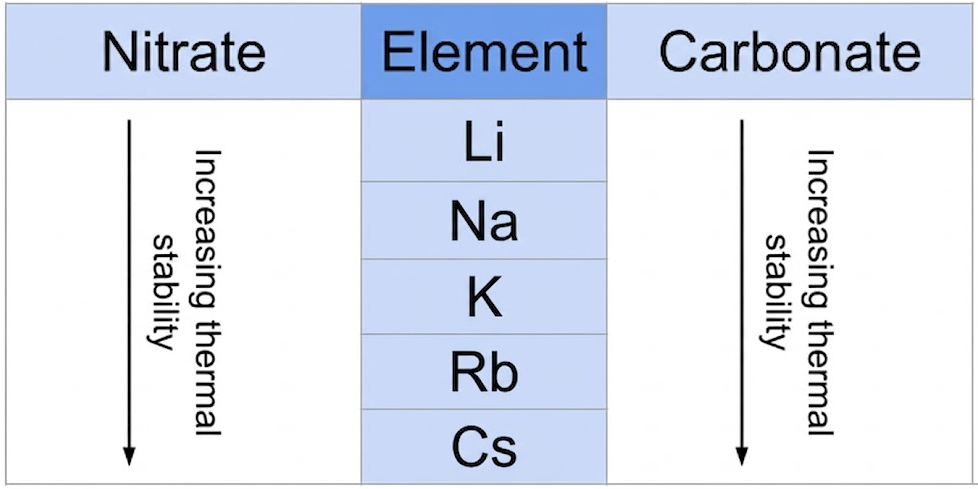
\( \mathrm{2MNO_3 \rightarrow 2MNO_2 + O_2} \)
Lithium (exception)
\( \mathrm{4LiNO_3 \rightarrow 2Li_2O + 4NO_2 + O_2} \)
Group 2 Nitrates
\( \mathrm{2M(NO_3)_2 \rightarrow 2MO + 4NO_2 + O_2} \)
Trend
- Stability increases down the group.
- Higher temperatures needed for decomposition.
Explanation
- Small cations (e.g. \( \mathrm{Li^+}, \mathrm{Mg^{2+}} \)) have high charge density.
- Strongly polarise \( \mathrm{CO_3^{2-}} \) or \( \mathrm{NO_3^-} \).
- Weakens internal bonds in anion.
- Easier decomposition → lower thermal stability.
- Larger cations (e.g. \( \mathrm{Ba^{2+}} \)) have lower charge density.
- Less polarisation → stronger anion bonds.
- Harder to decompose → higher thermal stability.
Group 1 vs Group 2
- Group 2 cations have +2 charge → higher polarising power.
- Therefore, Group 2 compounds are less thermally stable than Group 1 (for similar size).
Summary
- Thermal stability increases down both groups.
- Due to decreasing polarising power of cations.
- Smaller, highly charged cations destabilise anions more.
Therefore, the trend is explained by the relationship between cation size, charge density, and polarisation of the anion.
Example 1 :
Explain why magnesium carbonate decomposes at a lower temperature than barium carbonate.
▶️ Answer/Explanation
Mg\(^{2+}\) is smaller and has higher charge density.
It strongly polarises the carbonate ion.
Weakens bonds in \( \mathrm{CO_3^{2-}} \).
Therefore, decomposes more easily.
Example 2:
Explain why lithium nitrate decomposes to an oxide, while sodium nitrate forms a nitrite.
▶️ Answer/Explanation
Li\(^+\) is very small with high charge density.
Strongly polarises nitrate ion.
Leads to greater decomposition (forms oxide).
Na\(^+\) is larger and less polarising.
Therefore, forms nitrite instead.
