Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.17 Flame colours-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.17 Flame colours- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.17 Flame colours- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.17 understand the formation of characteristic flame colours by Group 1 and 2 compounds in terms of electron transitions
Students will be expected to know the flame colours for Group 1 and 2 compounds
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.17 Flame Colours and Electron Transitions
Group 1 and Group 2 metal compounds produce characteristic flame colours when heated. This is due to electron transitions in metal ions.
Electron Transitions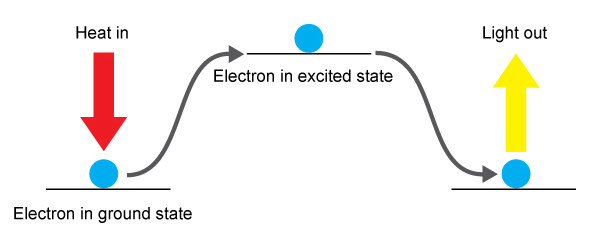
- Heat provides energy to electrons.
- Electrons are excited to higher energy levels.
- Electrons then fall back to lower energy levels.
- Energy is released as light (photons).
- The wavelength (colour) depends on energy gap.
Energy Relationship
\( \mathrm{\Delta E = h\nu} \)
- Larger energy gap → higher frequency → different colour.
Why Each Metal Gives a Different Colour
- Each metal ion has unique energy levels.
- Therefore, different energy gaps.
- Different wavelengths of light emitted.
Flame Colours to Know
Group 1

- \( \mathrm{Li^+} \) → crimson red
- \( \mathrm{Na^+} \) → yellow
- \( \mathrm{K^+} \) → lilac
Group 2
\( \mathrm{Mg^{2+}} \) → no characteristic colour (white flame)
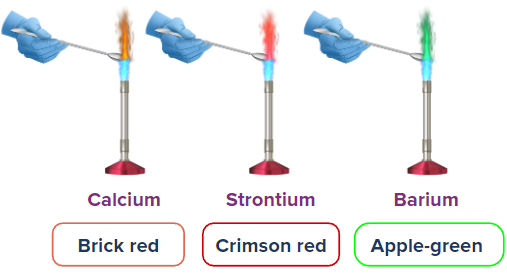
- \( \mathrm{Ca^{2+}} \) → brick red
- \( \mathrm{Sr^{2+}} \) → red
- \( \mathrm{Ba^{2+}} \) → apple green
Important Notes
- Sodium colour is very intense → can mask others.
- Flame tests are used for qualitative analysis.
Summary
- Flame colours arise from electron excitation and relaxation.
- Each metal has a unique emission spectrum.
- Specific colours must be memorised for exams.
Therefore, flame tests provide evidence for quantised energy levels in atoms.
Flame Colours of Group 1 and Group 2 Elements
| Group | Element / Ion | Flame Colour |
|---|---|---|
| Group 1 | \( \mathrm{Li^+} \) | Crimson red |
| Group 1 | \( \mathrm{Na^+} \) | Yellow |
| Group 1 | \( \mathrm{K^+} \) | Lilac |
| Group 2 | \( \mathrm{Mg^{2+}} \) | No characteristic colour (white) |
| Group 2 | \( \mathrm{Ca^{2+}} \) | Brick red |
| Group 2 | \( \mathrm{Sr^{2+}} \) | Red |
| Group 2 | \( \mathrm{Ba^{2+}} \) | Apple green |
Example 1 :
Explain why sodium produces a yellow flame in a flame test.
▶️ Answer/Explanation
Heat excites electrons in sodium atoms.
Electrons fall back to lower energy levels.
Energy released corresponds to yellow light.
Example 2 :
Explain why different Group 2 metals produce different flame colours.
▶️ Answer/Explanation
Each metal ion has different energy level spacings.
Electrons release different amounts of energy when returning.
Therefore, different wavelengths (colours) are emitted.
