Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.18 Thermal decomposition and flame tests-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.18 Thermal decomposition and flame tests- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.18 Thermal decomposition and flame tests- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.18 know experimental procedures to show:
i patterns in the thermal decomposition of Group 1 and 2 nitrates and carbonates
Students will be expected to know tests for carbon dioxide and oxygen; and to recognise nitrogen dioxide by its colour and acidic pH
ii flame colours in compounds of Group 1 and 2 elements
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.18 (i) Experimental Procedures: Thermal Decomposition of Group 1 & Group 2 Compounds
These experiments are used to observe patterns in thermal stability by heating nitrates and carbonates and identifying the gases produced.
(A) Thermal Decomposition of Carbonates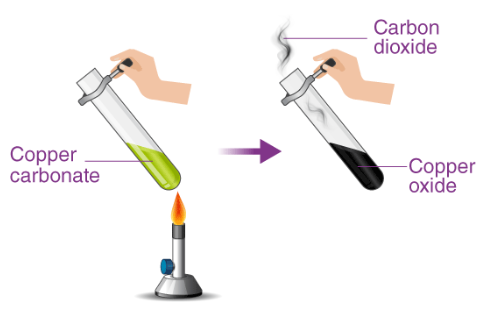
Procedure
- Place solid carbonate (e.g. \( \mathrm{CaCO_3} \)) in a test tube.
- Heat strongly using a Bunsen burner.
- Pass gas produced into limewater.
Observation
- Limewater turns milky/cloudy.
Conclusion
- Gas produced is \( \mathrm{CO_2} \).
\( \mathrm{MCO_3 \rightarrow MO + CO_2} \)
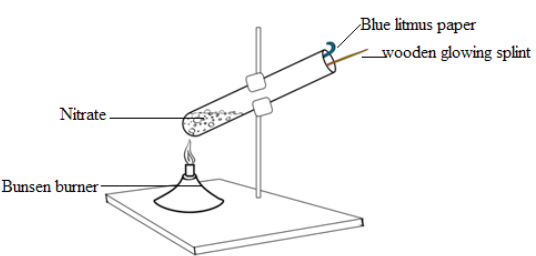
(B) Thermal Decomposition of Nitrates
Procedure
- Place metal nitrate (e.g. \( \mathrm{Mg(NO_3)_2} \)) in a test tube.

- Heat gently then strongly.
- Test gases evolved.
Observations & Tests
Oxygen (\( \mathrm{O_2} \)):
- Glowing splint relights.
Nitrogen dioxide (\( \mathrm{NO_2} \)):
- Brown gas.
- Turns damp indicator paper acidic (red).
Equations
Group 2 Nitrates
\( \mathrm{2M(NO_3)_2 \rightarrow 2MO + 4NO_2 + O_2} \)
Group 1 (except Li)
\( \mathrm{2MNO_3 \rightarrow 2MNO_2 + O_2} \)
Patterns Observed
- Down the group → more heat required.
- Shows increasing thermal stability.
Key Tests to Remember
- \( \mathrm{CO_2} \) → limewater turns milky
- \( \mathrm{O_2} \) → glowing splint relights
- \( \mathrm{NO_2} \) → brown gas + acidic
Summary
- Heating carbonates produces \( \mathrm{CO_2} \).
- Heating nitrates produces \( \mathrm{O_2} \) and \( \mathrm{NO_2} \) (Group 2).
- Observations confirm decomposition patterns and trends.
Therefore, these experiments provide evidence for thermal stability trends in Groups 1 and 2.
Example 1:
Describe how you would identify the gases produced when heating magnesium nitrate.
▶️ Answer/Explanation
Oxygen relights a glowing splint.
Nitrogen dioxide appears as a brown gas.
It turns damp indicator paper acidic.
8.18 (ii) Experimental Procedure: Flame Colours of Group 1 and Group 2 Compounds
Flame tests are used to identify metal ions based on the characteristic colour they produce when heated.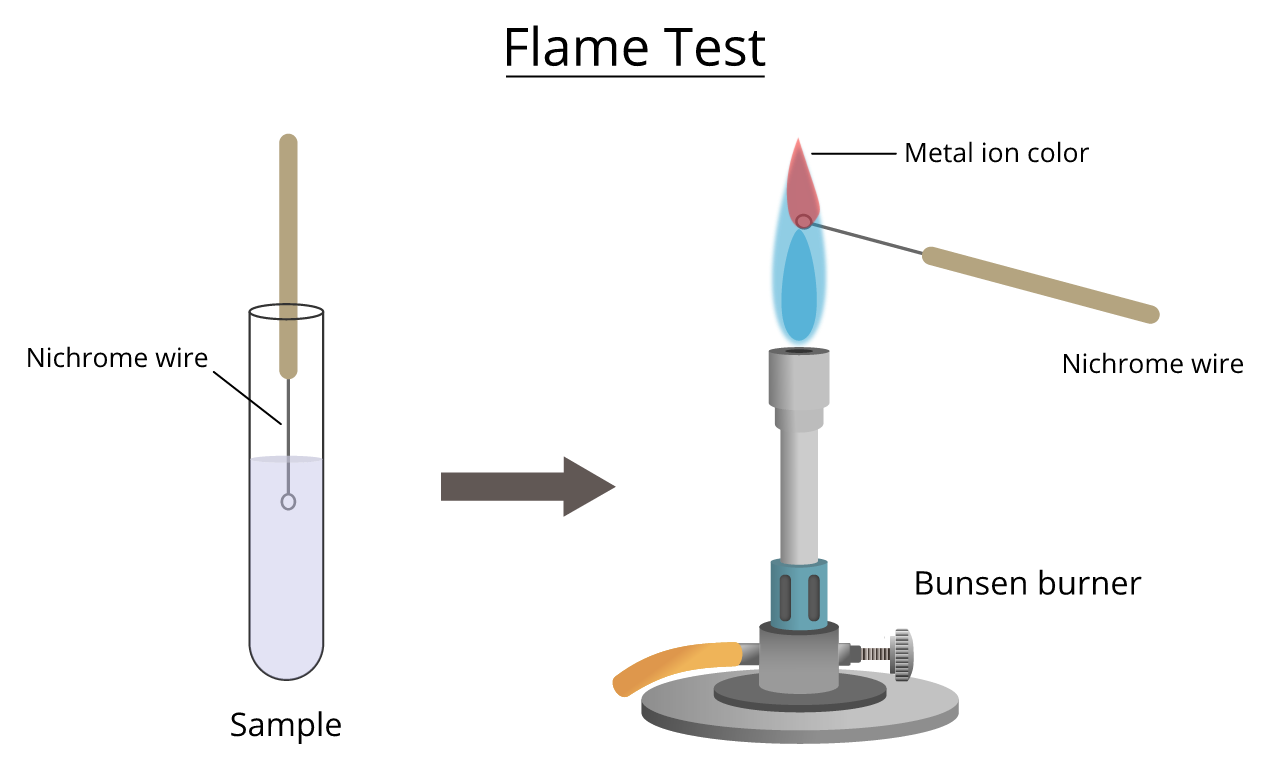
Apparatus
- Bunsen burner
- Platinum / nichrome wire loop
- Dilute \( \mathrm{HCl} \)
- Metal salt (solid or solution)
Procedure
- Clean the wire loop by dipping it in dilute \( \mathrm{HCl} \).
- Place it in the Bunsen flame until no colour is seen.
- Dip the clean loop into the sample.
- Place the loop in the blue (non-luminous) flame.
- Observe and record the flame colour.
Observations (Colours)
- \( \mathrm{Li^+} \) → crimson red
- \( \mathrm{Na^+} \) → yellow
- \( \mathrm{K^+} \) → lilac
- \( \mathrm{Ca^{2+}} \) → brick red
- \( \mathrm{Sr^{2+}} \) → red
- \( \mathrm{Ba^{2+}} \) → apple green
Explanation
- Heat excites electrons to higher energy levels.
- Electrons return to lower levels.
- Energy released as light of specific wavelength.
Precautions
- Always clean wire thoroughly to avoid contamination.
- Use blue flame (non-luminous) for clear observation.
- Sodium contamination may mask other colours.
Summary
- Flame tests identify metal ions by colour.
- Caused by electron transitions.
- Simple qualitative analysis method.
Therefore, flame tests provide quick identification of Group 1 and Group 2 ions.
Example 2:
Explain why it is important to clean the wire loop before each flame test.
▶️ Answer/Explanation
Residues from previous samples may remain.
This can produce incorrect flame colours.
Therefore, cleaning ensures accurate identification.