Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.19 Tests for CO₃²⁻, SO₄²⁻, NH₄⁺-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.19 Tests for CO₃²⁻, SO₄²⁻, NH₄⁺- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.19 Tests for CO₃²⁻, SO₄²⁻, NH₄⁺- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.19 know reactions, including ionic equations where appropriate, for identifying:
i carbonate ions, CO₃²⁻, and hydrogencarbonate ions, HCO₃⁻, using an aqueous acid to form carbon dioxide (and testing the gas with limewater)
ii sulfate ions, SO₄²⁻, using acidified barium chloride solution
iii ammonium ions, NH₄⁺, using sodium hydroxide solution and warming to form ammonia (and testing with litmus and HCl fumes)
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.19 Tests for Anions and Ammonium Ion
These are standard qualitative analysis tests used to identify specific ions using characteristic reactions and observations.
(i) Carbonate \( \mathrm{CO_3^{2-}} \) and Hydrogencarbonate \( \mathrm{HCO_3^-} \)
Reagent
- Dilute acid (e.g. \( \mathrm{HCl} \))
Observations
- Effervescence (bubbling) → gas produced
- Gas turns limewater milky
Conclusion
- Gas is \( \mathrm{CO_2} \)
Ionic Equations
\( \mathrm{CO_3^{2-} + 2H^+ \rightarrow CO_2 + H_2O} \)
\( \mathrm{HCO_3^- + H^+ \rightarrow CO_2 + H_2O} \)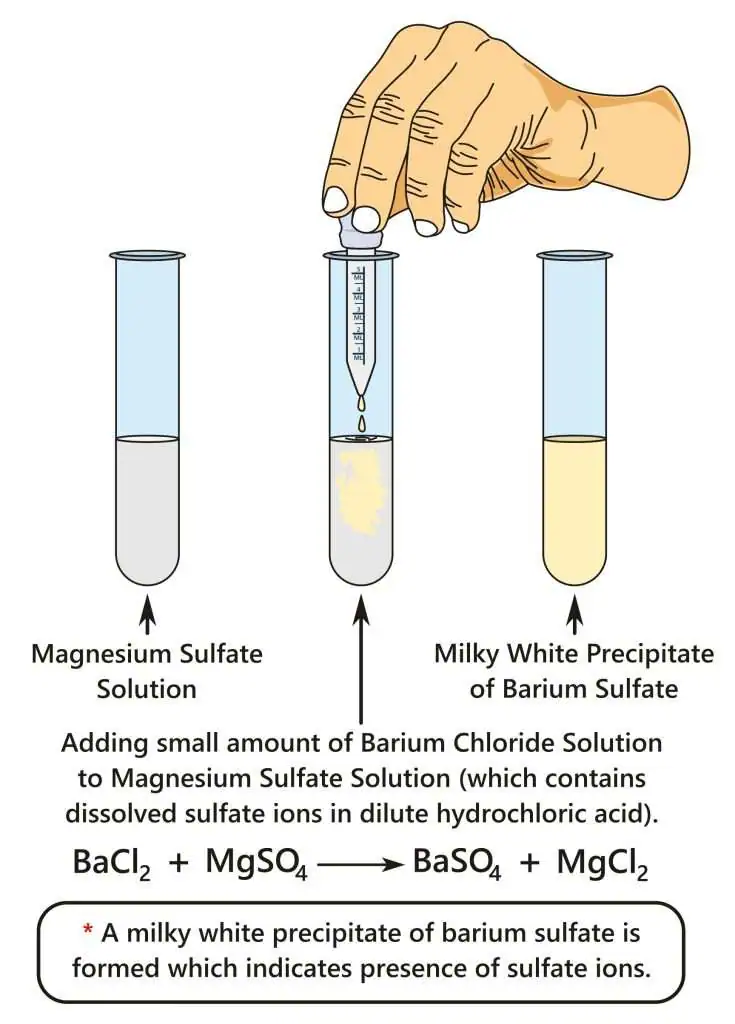
\( \mathrm{CO_2 + Ca(OH)_2 \rightarrow CaCO_3 + H_2O} \)
(ii) Sulfate \( \mathrm{SO_4^{2-}} \)
Reagent
- Acidified barium chloride solution (\( \mathrm{BaCl_2 + HCl} \))
Observation
- White precipitate forms
Conclusion
- Precipitate is \( \mathrm{BaSO_4} \) → sulfate present
Ionic Equation
\( \mathrm{Ba^{2+} + SO_4^{2-} \rightarrow BaSO_4(s)} \)
(iii) Ammonium \( \mathrm{NH_4^+} \)
Reagent
- Sodium hydroxide solution
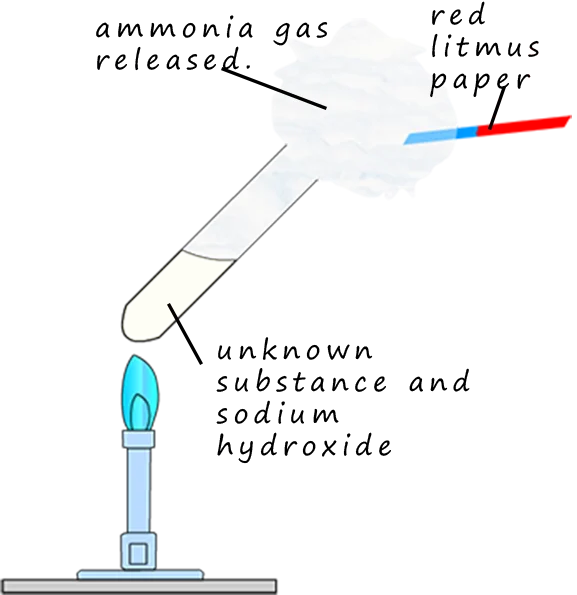
- Gentle heating
Observations
- Pungent gas (ammonia) evolved
- Damp red litmus turns blue
- White fumes with \( \mathrm{HCl} \) (ammonium chloride)
Ionic Equation
\( \mathrm{NH_4^+ + OH^- \rightarrow NH_3 + H_2O} \)
Summary Table
| Ion | Reagent | Observation |
|---|---|---|
| \( \mathrm{CO_3^{2-}} \) / \( \mathrm{HCO_3^-} \) | Dilute acid | Effervescence, limewater turns milky |
| \( \mathrm{SO_4^{2-}} \) | Acidified \( \mathrm{BaCl_2} \) | White precipitate |
| \( \mathrm{NH_4^+} \) | NaOH + heat | Ammonia gas, turns litmus blue, white fumes with HCl |
Summary
- Carbonates → \( \mathrm{CO_2} \) → limewater test
- Sulfates → white \( \mathrm{BaSO_4} \) precipitate
- Ammonium → \( \mathrm{NH_3} \) gas → litmus + HCl test
Therefore, these reactions allow reliable identification of key ions in qualitative analysis.
Example 1
A solid sample produces effervescence when dilute hydrochloric acid is added. The gas turns limewater milky. Identify the ion present and write the ionic equation.
▶️ Answer/Explanation
Ion: \( \mathrm{CO_3^{2-}} \) or \( \mathrm{HCO_3^-} \)
Reason: CO\(_2\) produced gives milky limewater
Ionic equation:
\( \mathrm{CO_3^{2-} + 2H^+ \rightarrow CO_2 + H_2O} \)
Example 2
A solution forms a white precipitate when acidified barium chloride is added. Explain why the solution must be acidified before adding barium chloride.
▶️ Answer/Explanation
Acid removes interfering ions such as carbonates.
Prevents formation of \( \mathrm{BaCO_3} \) (false positive).
Ensures precipitate is due to \( \mathrm{SO_4^{2-}} \) only.
Example 3
A compound is warmed with sodium hydroxide and produces a gas that turns damp red litmus paper blue and forms white fumes with hydrogen chloride. Identify the ion and write the ionic equation.
▶️ Answer/Explanation
Ion: \( \mathrm{NH_4^+} \)
Gas: \( \mathrm{NH_3} \)
Ionic equation:
\( \mathrm{NH_4^+ + OH^- \rightarrow NH_3 + H_2O} \)
