Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.20 Solution concentration and titrations-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.20 Solution concentration and titrations- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.20 Solution concentration and titrations- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.20 be able to calculate solution concentrations, in mol dm⁻³ and g dm⁻³, including simple acid-base titrations using the indicators methyl orange and phenolphthalein
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.20 Solution Concentrations & Acid–Base Titrations
This topic involves calculating concentrations in \( \mathrm{mol\ dm^{-3}} \) and \( \mathrm{g\ dm^{-3}} \), and applying these in acid–base titrations.
(A) Concentration Definitions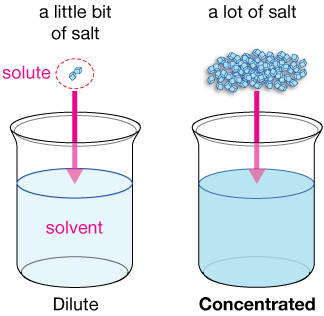
Molar concentration:
\( \mathrm{c = \frac{n}{V}} \)
- \( c \) = concentration (mol dm\(^{-3}\))
- \( n \) = moles (mol)
- \( V \) = volume (dm\(^3\))
Mass concentration:
\( \mathrm{c = \frac{mass}{volume}} \)
To convert:
\( \mathrm{c(g\ dm^{-3}) = c(mol\ dm^{-3}) \times M_r} \)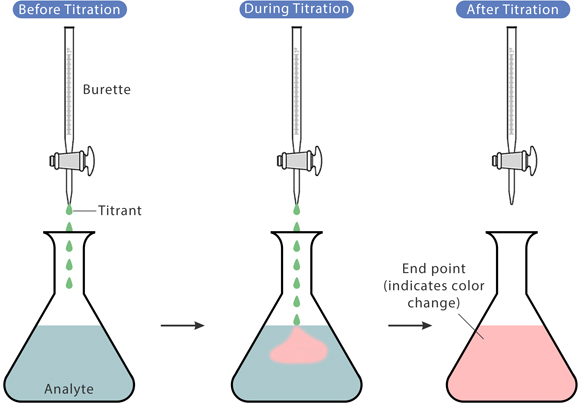
(B) Titration Principle
At equivalence point:
\( \mathrm{moles\ acid = moles\ base} \) (taking stoichiometry into account)
General Method
- Step 1: Write balanced equation
- Step 2: Calculate moles using \( \mathrm{n = cV} \)
- Step 3: Use mole ratio
- Step 4: Find unknown concentration
(C) Indicators
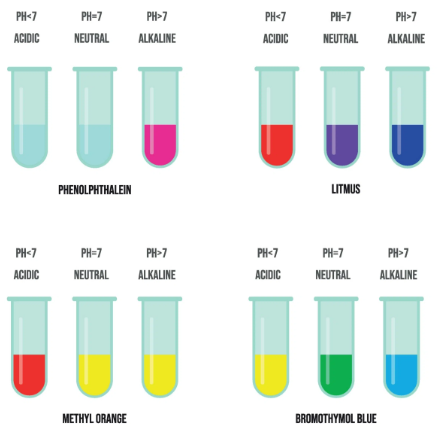
| Indicator | Colour Change | Use |
|---|---|---|
| Methyl orange | Red → Yellow | Strong acid + weak base |
| Phenolphthalein | Colourless → Pink | Weak acid + strong base |
(D) Key Equations
- \( \mathrm{n = cV} \) (V in dm\(^3\))
- \( \mathrm{c = \frac{n}{V}} \)
- \( \mathrm{n = \frac{mass}{M_r}} \)
Example 1
Calculate the concentration when 0.50 mol is dissolved in 250 cm\(^3\).
▶️ Answer/Explanation
Convert volume:
\( \mathrm{250\ cm^3 = 0.250\ dm^3} \)
\( \mathrm{c = \frac{0.50}{0.250} = 2.0\ mol\ dm^{-3}} \)
Example 2
Convert 1.5 mol dm\(^{-3}\) of \( \mathrm{NaCl} \) to g dm\(^{-3}\).
▶️ Answer/Explanation
\( M_r = 58.5 \)
\( \mathrm{c = 1.5 \times 58.5 = 87.75\ g\ dm^{-3}} \)
Example 3
25.0 cm\(^3\) of \( \mathrm{NaOH} \) reacts with 20.0 cm\(^3\) of 0.100 mol dm\(^{-3}\) \( \mathrm{HCl} \). Find the concentration of NaOH.
▶️ Answer/Explanation
Equation:
\( \mathrm{HCl + NaOH \rightarrow NaCl + H_2O} \)
Moles HCl:
\( \mathrm{0.100 \times 0.0200 = 0.00200\ mol} \)
Ratio = 1:1 → moles NaOH = 0.00200 mol
Concentration NaOH:
\( \mathrm{c = \frac{0.00200}{0.0250} = 0.0800\ mol\ dm^{-3}} \)
