Edexcel International A Level (IAL) Chemistry (YCH11) - Unit 2 - 8.21 CORE PRACTICAL 3: HCl concentration-Study Notes - New Syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.21 CORE PRACTICAL 3: HCl concentration- Study Notes- New syllabus
Edexcel International A Level (IAL) Chemistry (YCH11) -Unit 2 – 8.21 CORE PRACTICAL 3: HCl concentration- Study Notes -International A Level (IAL) Chemistry (YCH11) – per latest Syllabus.
Key Concepts:
8.21 CORE PRACTICAL 3
Finding the concentration of a solution of hydrochloric acid
Edexcel International A Level (IAL) Chemistry (YCH11) -Concise Summary Notes- All Topics
8.21 CORE PRACTICAL 3 – Finding the Concentration of Hydrochloric Acid
This practical determines the concentration of \( \mathrm{HCl} \) using a standard solution (usually \( \mathrm{Na_2CO_3} \) or \( \mathrm{NaOH} \)) by titration.
Principle
Unknown acid concentration is found using a reaction with known concentration.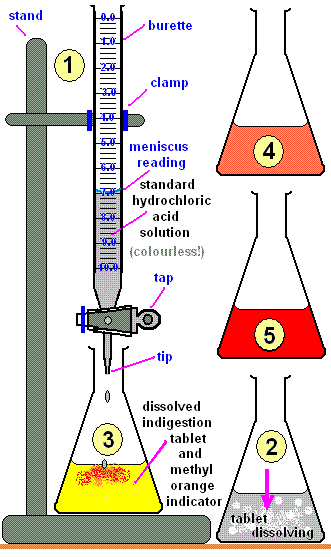
Typical Reaction (with Sodium Carbonate)
\( \mathrm{Na_2CO_3 + 2HCl \rightarrow 2NaCl + H_2O + CO_2} \)
Apparatus
- Burette (with \( \mathrm{HCl} \))
- Pipette (25.0 cm\(^3\))
- Conical flask
- Indicator (methyl orange)
- Standard solution (e.g. \( \mathrm{Na_2CO_3} \))
Procedure
- Use pipette to transfer 25.0 cm\(^3\) of standard solution into flask.
- Add a few drops of methyl orange.
- Fill burette with \( \mathrm{HCl} \).
- Add acid slowly while swirling.
- Stop at end point (colour change).
- Record titre. Repeat for concordant results.
Indicator Choice
- Methyl orange: yellow → orange/red
- Suitable for acid–carbonate reaction
Calculations
- Step 1: Calculate moles of standard solution
- Step 2: Use mole ratio (from equation)
- Step 3: Find moles of \( \mathrm{HCl} \)
- Step 4: Use \( \mathrm{c = \frac{n}{V}} \)
Key Experimental Skills
- Read burette at eye level (±0.05 cm\(^3\))
- Rinse apparatus with correct solutions
- Swirl continuously
- Add acid dropwise near endpoint
Sources of Error
- Overshooting endpoint
- Parallax error in reading burette
- Incomplete mixing
Summary
- Use titration to determine unknown \( \mathrm{HCl} \) concentration.
- Requires accurate measurements and correct indicator.
- Based on stoichiometry and \( \mathrm{n = cV} \).
Therefore, this practical links experimental technique with quantitative chemical analysis.
Example :
25.0 cm\(^3\) of 0.0500 mol dm\(^{-3}\) \( \mathrm{Na_2CO_3} \) requires 20.0 cm\(^3\) of \( \mathrm{HCl} \). Find the concentration of \( \mathrm{HCl} \).
▶️ Answer/Explanation
Moles Na\(_2\)CO\(_3\):
\( \mathrm{0.0500 \times 0.0250 = 0.00125\ mol} \)
Ratio: 1 : 2
Moles HCl:
\( \mathrm{0.00125 \times 2 = 0.00250\ mol} \)
Concentration:
\( \mathrm{c = \frac{0.00250}{0.0200} = 0.125\ mol\ dm^{-3}} \)
